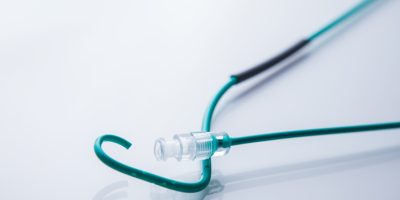
Penumbra JET 7 Xtra Flex Catheter Lawsuits
In December 2020, Penumbra issued a recall for its JET 7 Xtra Flex Catheter because of device malfunctions resulting in serious injury or death. Attorneys were investigating JET 7 Xtra Flex Catheter lawsuits from stroke patients claiming injuries from the device while having a blood clot removed. Drugwatch's legal partners are not accepting cases.
Our content is developed and backed by respected legal, medical and scientific experts. More than 30 contributors, including product liability attorneys and board-certified physicians, have reviewed our website to ensure it’s medically sound and legally accurate.
legal help when you need it most.
Drugwatch has provided people injured by harmful drugs and devices with reliable answers and experienced legal help since 2009. Brought to you by The Wilson Firm LLP, we've pursued justice for more than 20,000 families and secured $324 million in settlements and verdicts against negligent manufacturers.
More than 30 contributors, including mass tort attorneys and board-certified doctors, have reviewed our website and added their unique perspectives to ensure you get the most updated and highest quality information.
Drugwatch.com is AACI-certified as a trusted medical content website and is produced by lawyers, a patient advocate and award-winning journalists whose affiliations include the American Bar Association and the American Medical Writers Association.
About Drugwatch.com
- 17 Years of Advocacy
- $324 Million Recovered for Clients
- 20,000 Families Helped
- A+ BBB Rating
- 4.9 Stars from Google Reviews
Testimonials
I found Drugwatch to be very helpful with finding the right lawyers. We had the opportunity to share our story as well, so that more people can be aware of NEC. We are forever grateful for them.
- Legally reviewed by Christopher Edison, Esquire
- Last update: May 8, 2026
- Est. Read Time: 3 min read
Penumbra’s urgent voluntary recall applies to all configurations of the Penumbra JET 7 Reperfusion Catheter with Xtra Flex Technology (JET 7 Xtra Flex).
Doctors use these devices to remove blood clots and restore blood flow in people who have suffered an acute ischemic stroke. The catheter enters the body through a blood vessel in the leg and travels up through the neck. Once the catheter makes contact with the blood clot causing the stroke, doctors are able to vacuum out the clot and restore blood flow.
These reports included mentions of device malfunctions, causing serious injuries, including 14 patient deaths caused by distal tip damage and expansion and rupture following contrast injection.
As of June 2026, Drugwatch is not aware of any new updates in this litigation or how many lawsuits have been filed. There may be some law firms still accepting cases, but Drugwatch’s legal partners are currently not accepting cases.
Reasons for the Recall
Penumbra issued its urgent voluntary recall because of device malfunctions and breakage that could cause injuries and death.
“Penumbra is recalling the JET 7 Xtra Flex because the catheter may become susceptible to distal tip damage during use. Distal tip damage in conjunction with pressurization or contrast injection may result in potential vessel damage, and subsequent patient injury or death,” according to Penumbra’s Urgent Voluntary Medical Device Recall Notification.
Device failure reports include “ballooning, expansion, rupture, breakage or complete separation, and exposure of internal support coils near the distal tip region of the JET 7 Xtra Flex catheter” according to the U.S. Food and Drug Administration.
The FDA has provided 510(k) premarket notification numbers for the recalled devices.
FDA Actions and Label Warnings
After the clearance of the JET 7 Xtra Flex, the FDA received reports of device failure. The agency prompted Penumbra to issue a notification to health care providers with warnings and precautions to mitigate risks, according to the FDA’s recall notification.
On July 27, 2020, Penumbra sent a letter to health care providers with warnings.
“JET 7 Xtra Flex may become susceptible to expansion or rupture during contrast injection due to distal tip weakening from manipulation against resistance or use with other manufacturers’ revascularization devices. Distal tip expansion or rupture may cause vessel damage and subsequent patient injury or death,” the letter said.
The letter warned providers not to inject contrast media through the JET 7 Xtra Flex. Instead, they should use the guide catheter to inject contrast.
The labeling update did not stop reports of problems with the device. Some reports were related to providers not following instructions. The FDA requested that Penumbra remove all JET 7 Extra Flex from the market to protect patients.
Injuries Claimed in Lawsuits
Penumbra JET 7 Reperfusion Catheters with Xtra Flex Technology are associated with several potential side effects and complications. These include allergic reactions from contrast media, device malfunction, death, air embolism, hemorrhage at access site and infection.
Injuries in lawsuits are linked to device malfunction and breakage.
- Death
- Hemorrhage
- Stroke (cerebral infarction)
- Blood vessel damage
What to Do If You’ve Been Injured
The FDA recommends reporting the injury to MedWatch, the FDA Safety Information and Adverse Event Reporting program. This allows the FDA to identify risks with medical devices.
Make sure you discuss the injuries with your medical provider and understand what your options are for treatment.
Calling this number connects you with a Drugwatch.com representative. We will direct you to one of our trusted legal partners for a free case review.
Drugwatch.com's trusted legal partners support the organization's mission to keep people safe from dangerous drugs and medical devices. For more information, visit our partners page.