Prescription by Algorithm: The Ethical and Legal Questions Behind GLP-1 Advertising
An influencer pitch for a GLP-1 weight-loss drug revealed a troubling shift in how prescriptions are marketed and obtained online. As telehealth platforms, social media and algorithms reshape access, medical oversight may be taking a backseat, raising urgent ethical, legal and patient safety concerns.

A few weeks ago, I received an email that stopped me in my tracks.
A telehealth company contacted me with an offer. They asked me to promote their GLP-1 weight-loss program to my online followers.
The pitch was similar to many I get: Make content, share my experience and include an affiliate link.
But this time, the product was different — it was a prescription drug.
As an attorney who has worked on many product liability cases and spent years focused on consumer safety, I immediately saw warning signs in that email.
Feeling both curious and uneasy, I began to look into how these medications are marketed online. I discovered a whole network of influencers, telehealth startups and viral weight-loss stories centered on drugs like Ozempic and Wegovy.
On social media, prescription drugs are increasingly presented like lifestyle products. Influencers post dramatic before-and-after photos. Some share affiliate codes for telehealth sites that promise fast prescriptions after a short online quiz.
What used to be a careful discussion between patient and doctor is now often handled through marketing funnels and online platforms.
The troubling reality is that many of these prescriptions appear to be generated not by a doctor’s careful evaluation, but by a targeted algorithm.

The GLP-1 Gold Rush
GLP-1 receptor agonists have quickly become some of the most profitable drugs in pharmaceutical history.
These drugs were first made to treat Type 2 diabetes. They copy a natural hormone that helps control blood sugar and appetite. By slowing down digestion and making people feel full longer, they can lead to significant weight loss.
For many people, these drugs really do help. Clinical trials show that some users lost 15% or more of their body weight.
But the rapid success of these drugs has also created a huge business opportunity. Telehealth companies, wellness startups and online pharmacies are all rushing to meet the demand.
Social media is now one of the main ways these companies market their products.
Instead of using traditional ads, many companies now hire influencers to share their weight-loss stories and promote telehealth prescription services.
Sometimes, these promotions look almost identical to ads for beauty products or fitness supplements. Except that the product being sold is a powerful prescription medication that can carry significant risks.

My Experience With a Telehealth Prescription
To see how these platforms work, I decided to try one myself.
I went to a telehealth website advertising GLP-1 drugs and started signing up. The first page asked for basic details: name, email, age and location.
The next step was a brief medical questionnaire.
They asked about my height, weight and how much weight I wanted to lose. There were also a few general questions about my medical history.
To test the system, I entered exaggerated numbers. I input a height and weight that would categorize someone as already underweight on the BMI scale. Then I set a weight-loss goal that would be extremely unhealthy by any medical standard.
Within seconds, I was moved to the next screen.
“Great news! You’re pre-approved.”
The site recommended semaglutide for $299 per month or tirzepatide for $399 per month.
There were no follow-up questions about my unrealistic weight-loss goal, no request for medical records and no request for lab work.
Instead, the site suggested an extra purchase. For another $30 a month, I could buy medication for nausea to help with possible side effects.
I continued through the process until I reached the payment screen.
At that point, I was told that after I paid, a doctor would review my information, and my medication would ship within two days.
At no time did I have to schedule a meeting with a doctor. There was no virtual visit, no blood tests and no check of my medical history.
The whole process took under 10 minutes. What started as curiosity left me increasingly uneasy.
In some parts of the internet, it seems you only need a credit card to get powerful metabolic drugs.

The Complicated Reality Behind GLP-1 Drugs
Part of the confusion surrounding these medications comes from the difference between brand-name drugs and the growing market for compounded versions.
Brand-name GLP-1 drugs such as Ozempic, Wegovy and Mounjaro are manufactured by Novo Nordisk and Eli Lilly and Company, and approved by the U.S. Food and Drug Administration for specific medical conditions.
These medications have gone through clinical trials and regulatory review. Even so, they are not without controversy.
Federal regulators recently warned Novo Nordisk for failing to properly report potential side effects related to semaglutide. That warning followed increasing scrutiny of the drug’s safety profile.
Patients have also filed GLP-1 lawsuits alleging severe gastrointestinal injuries, including stomach paralysis, after using the drugs.
Other lawsuits center on vision loss in those taking GLP-1s. Recent studies examining patients taking semaglutide found that those using the drug had significantly higher odds of developing nonarteritic anterior ischemic optic neuropathy, or NAION, a rare condition that can cause sudden vision loss.
Emerging research is raising new concerns about the effects of these drugs on muscle and bone health. Because GLP-1 medications can lead to rapid weight loss, some patients may lose significant lean muscle mass during treatment.
These risks do not mean the medications are never appropriate. For many patients with obesity or diabetes, the benefits may be significant. But they underscore the importance of careful medical oversight.

The Rise of Compounded GLP-1 Drugs
If brand-name drugs come with risks, the situation becomes even murkier when compounded medications enter the picture.
Compounded drugs are custom medications prepared by pharmacies to meet specific patient needs. Under federal law, compounding can be appropriate when a patient requires a different dosage or formulation from what is commercially available.
Compounded drugs are not approved by the FDA and are not required to undergo the same safety and effectiveness testing as brand-name medications.
I spoke with Louiza Tarassova, a Florida attorney who handles med spa injury cases. She told me a major concern with compounded drugs is “the lack of reliability around dose, formulation and source.”
She also explained that “these unapproved products do not come with warning labels that advise their providers of the risks of taking the drug.” This raises questions over true informed consent for patients.
During shortages of GLP-1 drugs, many telehealth companies began offering compounded semaglutide or tirzepatide as alternatives.
In some cases, practitioners argue that compounded medications are tailored to individual patients, thereby allowing them to legally prescribe them.
But regulators are increasingly concerned about how these drugs are being marketed.
In March 2026, the FDA sent warning letters to 30 telehealth companies for illegally marketing compounded GLP-1 drugs. Regulators said many of the companies were promoting compounded medications as if they were equivalent to approved brand-name drugs.
The agency also warned companies about misleading claims about safety and effectiveness.
For most individuals scrolling through social media advertisements, distinguishing an FDA-approved drug from a compounded product can be almost impossible.
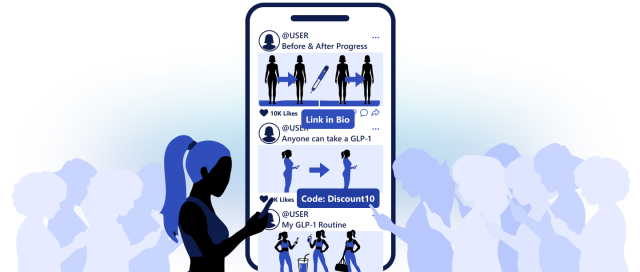
Influencers and Celebrities Fuel the GLP-1 Boom
Much of the demand for GLP-1 drugs now comes from influencer marketing.
If you scroll through social media, you will quickly find weight-loss stories linked to telehealth platforms. Influencers share their progress, show weekly injections and promote discount codes for prescription programs.
Once you show even slight interest in a post about GLP-1s, the algorithm ensures you continue to see that type of content.
Celebrities are also becoming a larger part of this marketing world.
Tennis champion Serena Williams has publicly discussed her use of GLP-1 medications and has partnered with a health company that connects consumers with telehealth weight-loss services. Her husband is an investor in the company and serves on its board.
Similarly, reality television personality Savannah Chrisley has also promoted GLP-1 medications through a wellness brand she owns.
The line between personal stories, ads and medical advice can get blurry in these spaces. For consumers, it is hard to tell the difference between real medical advice and paid promotion.

How Is This Legal?
The United States already has one of the most permissive pharmaceutical advertising systems in the world. Only the United States and New Zealand allow direct-to-consumer prescription drug advertising.
But social media has created a regulatory gray area.
Kickbacks to physicians for prescribing drugs are illegal under federal law. Pharmaceutical companies are heavily restricted in how they compensate doctors for promoting medications.
Influencers, however, fall into a different category. They are treated as marketers.
Under Federal Trade Commission rules, influencers must disclose when they are paid to promote products. In reality, those disclosures are often nonexistent, buried in hashtags or difficult to see.
The result is a marketing environment in which prescription drugs are promoted through testimonials, affiliate links and transformation stories that blur the line between personal experience and advertising.
Adding to the concerns, there is a growing market for compounded versions of GLP-1 drugs and peptides sold online. Some of these products are labeled “research chemicals” and marked “not for human consumption,” yet are openly marketed to people seeking weight loss.
In many cases, consumers have little idea where the drugs are manufactured or whether they have been evaluated for safety.
In some cases, they are not even sure whether they are receiving an FDA-approved drug or a compounded alternative.

The Return of Thin at Any Cost
Spend a few minutes scrolling through TikTok or Instagram, and a clear pattern emerges.
Many GLP-1 promotions target women who are not medically obese or diabetic. The messaging often suggests these drugs are for anyone who wants to lose a few stubborn pounds.
As soon as I started researching this topic, I began receiving retargeted ads from several telehealth websites I had visited. Some of the ads were shocking.
“You don’t have to be obese to try a GLP-1.”
“Lose the last ten pounds.”
“Get your summer body faster.”
Many videos feature women who are already thin explaining they only want to lose a few pounds. A doctor reassures them these medications are perfect for that goal.
Many advertisements also promote the drugs as effortless weight loss. Some influencers say they can eat whatever they want. Others claim they no longer need exercise.
The message is simple: Take this shot and the weight will disappear.
Health experts are increasingly warning that the reality is much more complicated.
Because GLP-1 medications can reduce appetite so dramatically, patients are often advised to prioritize adequate protein intake and engage in regular strength training while using the drugs. Maintaining muscle mass and proper nutrition is critical to preventing unhealthy muscle loss during rapid weight reduction.
But you rarely see that advice in influencer ads.
Instead of emphasizing careful medical monitoring, balanced nutrition and exercise, many marketing campaigns present the drugs as a shortcut to thinness. For women who grew up during the peak of extreme diet culture, this messaging feels very familiar.
The difference now is that instead of detox teas or appetite suppressants, the product promoted is a prescription medication that alters the body’s metabolic system.
Eating disorder specialists have already raised concerns about the potential impact of GLP-1 drugs on people with a history of disordered eating.
“No one is screening for disordered eating before prescribing,” said Deanna Wolfe, a registered dietitian who has helped hundreds of women recover from disordered eating. “GLP-1s can be helpful for some people and extremely harmful for this population. Many women are hopping on these drugs to lose weight to reach unrealistic standards that we have right now in Hollywood.”
Normalizing rapid weight loss through medication could reinforce harmful ideas about body image and food restriction. Unlike traditional dieting trends, these drugs come with medical risks that are rarely discussed in influencer videos.

The Ethical Questions
GLP-1 drugs represent a genuine medical breakthrough for many people struggling with obesity and metabolic disease. But the way they are marketed raises uncomfortable questions about the future of health care.
Should influencers and celebrities be promoting prescription drugs?
Should telehealth companies be allowed to recruit creators to advertise medications through affiliate links?
And what happens when the algorithms that drive social media engagement also start driving demand for pharmaceuticals?
The email I received was probably not unusual. Thousands of creators are likely receiving similar offers every day.
But when prescriptions are routed through digital questionnaires, marketing funnels and affiliate links, the traditional role of the physician begins to fade into the background.
In today’s GLP-1 boom, more and more prescriptions are beginning not inside a doctor’s office, but inside an algorithm.
Calling this number connects you with a Drugwatch.com representative. We will direct you to one of our trusted legal partners for a free case review.
Drugwatch.com's trusted legal partners support the organization's mission to keep people safe from dangerous drugs and medical devices. For more information, visit our partners page.




