Judge Denies Request To Delay Bard PowerPort Lawsuit Trials
A U.S. District judge in Arizona has chosen to proceed with the bellwether trials for the Bard PowerPort multidistrict litigation over objections from the manufacturer.
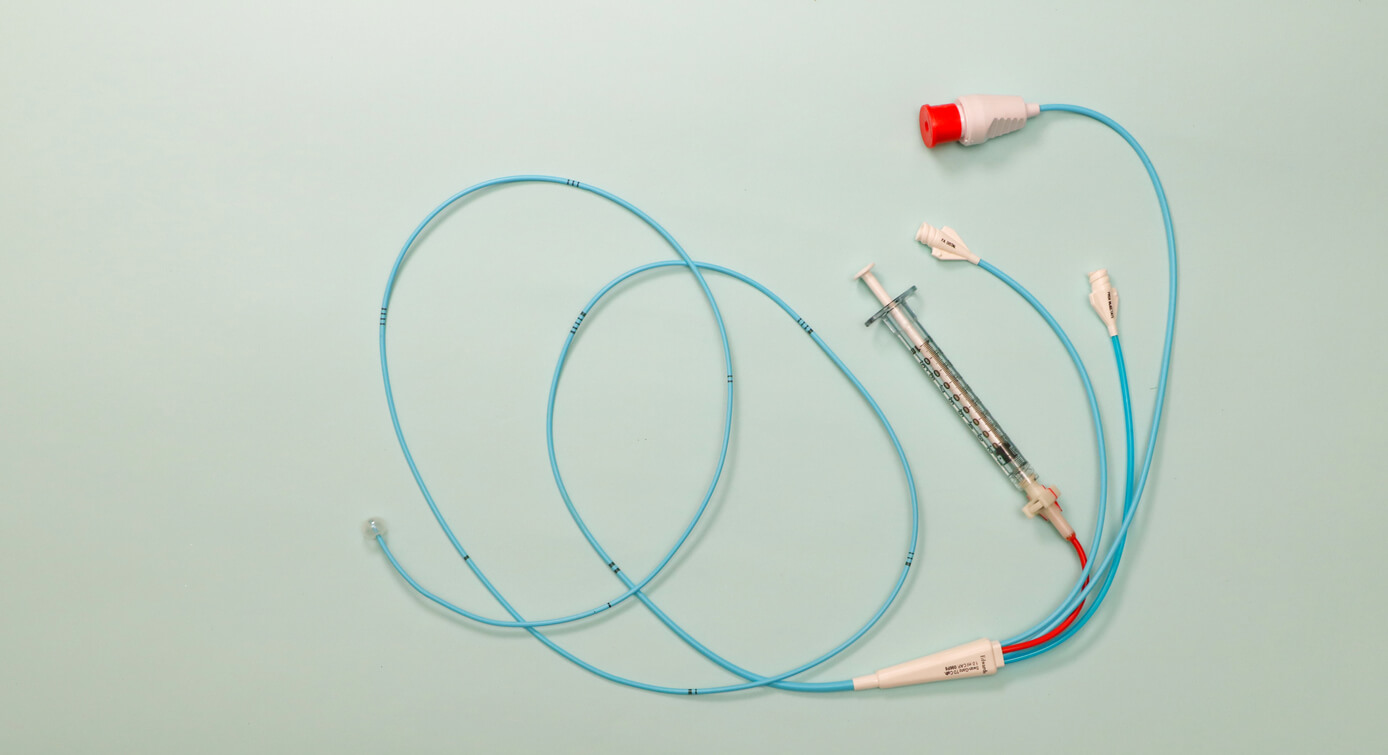
As of April 1, 2024, C.R. Bard had 116 pending PowerPort lawsuits in Judge David G. Campbell’s federal courtroom. Experts expect that number to grow. The PowerPort lawsuits claim the implanted port catheters caused severe complications, such as infections and blood clots.
Campbell opted not to delay selecting representative cases for early bellwether trial dates. Bellwether trials are test trials representing dozens or hundreds of similar lawsuits. The outcome of bellwether trials in an MDL can determine whether plaintiffs stick with their lawsuits or whether the defendant agrees to a settlement.
The judge’s order for the bellwether trial process to proceed sets the stage for bellwether cases affecting thousands of individuals claiming Bard PowerPort complications.
Number of Lawsuits Expected To Grow
Campbell rejected Bard’s request to delay the bellwether trial selections in the Bard PowerPort MDL, emphasizing the importance of maintaining the current schedule. He instructed attorneys from both sides to choose bellwether candidates by July 1, 2024.
The judge’s order for the bellwether trial process to proceed sets the stage for cases potentially affecting thousands of individuals claiming Bard PowerPort complications.
Drawing from experience with Bard IVC filter lawsuits, where case numbers grew significantly over time, the judge expressed skepticism that the current case count accurately reflected the eventual total. He highlighted the potential for a substantial increase in filings, as plaintiffs’ lawyers anticipate over 2,000 lawsuits.
Lawsuits Claim Wide Range of Serious Complications
Lawsuits filed against C.R. Bard and its parent company, Becton Dickinson, claim PowerPort and other Bard chemo devices pose potential risks such as fracturing, migration and infections. Despite these claims, Bard marketed the device as safe. Federal courts consolidated all lawsuits over Bard chemo port devices into an MDL in August 2023.
Typical lawsuits in the Bard PowerPort MDL involve patients who suffered severe complications from the implanted devices. Brandie Songy of Texas, for example, claims Bard knew about the risks in one case but continued to market the PowerPort as safe. Her lawsuit claims injuries ranging from cardiac arrhythmia to severe pain.
In another lawsuit, Johanna Randow of Maryland accuses Bard of negligence after her Bard Infuse-A-Port fractured, sending fragments into her heart and causing a pulmonary embolism. Despite having the port removed, Randow suffered complications years later.
These legal complaints against Bard claim a pattern of device faults, including tubing issues leading to bacterial buildup and plastic fragments causing heart problems.




