Benzocaine: Side Effects, FDA Warnings & Lawsuit Information
Benzocaine is a topical anesthetic in sprays, gels and oral products. It relieves pain but carries serious risks like a rare but life-threatening blood disorder. The FDA has issued warnings, and lawsuits have been filed claiming that manufacturers failed to warn people about potential risks.
Our content is developed and backed by respected legal, medical and scientific experts. More than 30 contributors, including product liability attorneys and board-certified physicians, have reviewed our website to ensure it’s medically sound and legally accurate.
legal help when you need it most.
Drugwatch has provided people injured by harmful drugs and devices with reliable answers and experienced legal help since 2009. Brought to you by The Wilson Firm LLP, we've pursued justice for more than 20,000 families and secured $324 million in settlements and verdicts against negligent manufacturers.
More than 30 contributors, including mass tort attorneys and board-certified doctors, have reviewed our website and added their unique perspectives to ensure you get the most updated and highest quality information.
Drugwatch.com is AACI-certified as a trusted medical content website and is produced by lawyers, a patient advocate and award-winning journalists whose affiliations include the American Bar Association and the American Medical Writers Association.
About Drugwatch.com
- 15+ Years of Advocacy
- $324 Million Recovered for Clients
- 20,000 Families Helped
- A+ BBB Rating
- 4.9 Stars from Google Reviews
Testimonials
I found Drugwatch to be very helpful with finding the right lawyers. We had the opportunity to share our story as well, so that more people can be aware of NEC. We are forever grateful for them.
- Medically reviewed by Mireille Hobeika, Pharm.D.
- Last update: May 6, 2026
- Est. Read Time: 5 min read
What Is Benzocaine Used For?
Benzocaine is a local anesthetic found in gels, lozenges and sprays. It’s often used orally to treat conditions like toothaches, sore throats, canker sores and gum discomfort.
As a topical application, it can help alleviate pain associated with minor skin issues, including sunburns and insect bites. Doctors also use benzocaine during dental procedures and before anesthesia.
Benzocaine is similar to another local anesthetic called lidocaine. While lidocaine also aids with pain relief, benzocaine is less potent and shorter-acting. Lidocaine is stronger and can be used for local anesthesia in medical procedures or administered intravenously for intubated patients.
Benzocaine Gels, Sprays & Menthol Products
Benzocaine is available in several over-the-counter forms, including gels for oral pain relief and sprays for sore throats. However, the FDA reports that benzocaine sprays may increase your risk of blood oxygen issues when used for medical procedures.
Another option is benzocaine menthol. This combines the anesthetic with menthol, an organic compound that provides a cooling sensation, to create soothing lozenges for sore throats.
Benzocaine Side Effects
People who use benzocaine often report minor side effects like local irritation and tingling. Benzocaine users may also experience serious risks that could be life-threatening, including allergic reactions, heart issues and a rare blood disorder called methemoglobinemia.
As of November 1, 2025, the Food and Drug Administration’s (FDA) Adverse Events Reporting System (FAERS) had received over 1,400 adverse event reports related to Benzocaine. Roughly 75% of these cases were serious.
| FDA Adverse Event Reports for Benzocaine Side Effects | |
|---|---|
| Total cases reported | 1,423 |
| Serious cases (including deaths) | 1,069 |
| Deaths | 58 |
Disclaimer: Reports sent to the FDA don’t necessarily mean the drug caused an adverse event. Consult a health care professional before stopping or changing medication.
Common Benzocaine Side Effects
Localized skin or application-site reactions are frequently reported by benzocaine users. These side effects are typically mild.
- Burning
- Flaky skin
- Itchiness
- Red skin
- Skin tenderness
- Tingling in your mouth
If you experience any side effects while taking benzocaine, report them to your doctor.
Serious Benzocaine Side Effects
The most serious benzocaine side effects include allergic reactions, heart rate changes and a rare but life-threatening blood disorder called methemoglobinemia.
- Allergic reactions, including facial swelling, skin irritation, hoarseness and difficulty speaking or breathing
- Changes in heart rhythm, including chest pain, irregular or fast heartbeat, difficulty breathing, dizziness and feeling lightheaded
- Methemoglobinemia
Severe allergic reactions to benzocaine can cause anaphylaxis, which is sometimes fatal. It may also lead to contact dermatitis, which can cause your skin to slough off and become infected.
Notify your doctor if you notice symptoms like difficulty breathing, changes in your heart rate, swelling or skin irritation. These could indicate a serious health condition requiring medical attention.
Benzocaine and Methemoglobinemia
Some benzocaine users develop a potentially fatal complication called methemoglobinemia. This condition causes elevated levels of methemoglobin in your blood.
Methemoglobin is a type of protein that cannot bind to oxygen. Excess levels reduce the amount of oxygen carried through your blood.
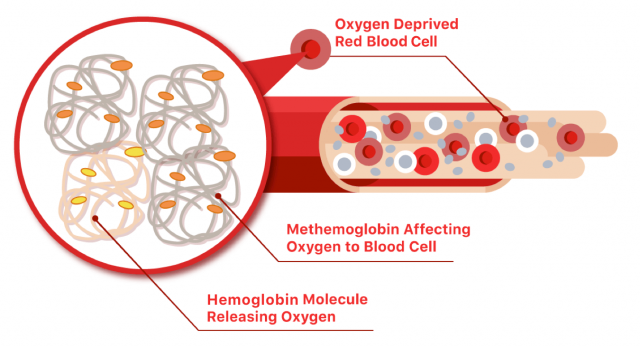
- Cyanosis (pale, gray or blue-colored skin)
- Dizziness
- Fatigue
- Headache
- Rapid heart rate
- Shortness of breath
Seek medical attention if you notice symptoms consistent with methemoglobinemia. These symptoms worsen as methemoglobin levels increase in the blood. If left untreated, the condition can cause coma or death.
Case Study: Benzocaine Throat Spray and Methemoglobinemia
After undergoing stomach surgery, a 46-year-old woman used a benzocaine throat spray to relieve her pain. However, she used too much of the spray over four days, resulting in an overdose. A case report in the Journal of Pharmacy and Therapeutics said this caused her to have breathing problems, and her skin turned blue.
Doctors diagnosed her condition as methemoglobinemia. This condition makes it difficult for the blood to carry oxygen.
Her methemoglobin levels were extremely high at 38.8%. These levels should typically be under 1%.
Doctors treated her with methylene blue, reversing the methemoglobinemia. Her oxygen levels returned to normal after receiving two doses of the treatment.
The authors said the case highlighted the dangers of overusing benzocaine products. They noted that methemoglobinemia requires prompt intervention to prevent severe harm. Following dosage guidelines closely is important to avoid dangerous side effects.
“Clinicians have long pointed out that ambiguous package instructions for use of the spray canisters of benzocaine products can be easily misinterpreted and can lead to potential overdoses.”
FDA Warnings and Recalls
In 2018, the U.S. Food and Drug Administration (FDA) issued a warning that disclosed a link between benzocaine and methemoglobinemia in infants and children younger than two years. The FDA advised against over-the-counter oral benzocaine products being used in children under the age of two.
“The FDA is warning that over-the-counter (OTC) oral drug products containing benzocaine should not be used to treat infants and children younger than 2 years ... These products carry serious risks and provide little to no benefit for the treatment of oral pain, including sore gums in infants due to teething.”
The FDA also announced a stronger warning about the risk of developing methemoglobinemia when using benzocaine. The agency estimated that over 400 patients developed methemoglobinemia after using benzocaine from February 2009 and October 2017.
In December 2023, Insight Pharmaceuticals recalled one lot of its Americaine 20% Benzocaine Topical Anesthetic Spray. This recall was due to the detection of benzene, a carcinogenic chemical. No serious adverse events were reported for this recalled product.
Benzocaine vs. Lidocaine
Benzocaine and lidocaine are both local anesthetics that have a numbing, pain-relieving effect. While lidocaine is more potent and longer-lasting than benzocaine, both medications share some common applications and risks.
Here’s how benzocaine and lidocaine compare.
| Benzocaine | Lidocaine | |
|---|---|---|
| Uses | Pain relief in your mouth and throat due to toothache, canker sores, painful gums, sore throat, and minor dental work like braces and dentures. Treating hemorrhoids and rectal irritation. Relieving itching or burning on the skin due to sunburn, insect bites, and other mild irritants. | Oral applications include reducing gagging during dental procedures and relieving pain from a sore mouth or throat. Providing local anesthetic during medical procedures. Relieving pain from burns, cuts, scratches, nerve pain, insect bites, and poison oak/ivy/sumac. Lubricating the throat, nose, or mouth before intubation. Treating urethritis and other urethral-related issues. |
| Strength | Weaker | Stronger |
| Duration | ~10 minutes | ~90 minutes |
| Risks | Methemoglobinemia, allergic reactions and heart problems | Methemoglobinemia and allergic reactions (all applications). Toxic reactions, choking, and difficulty swallowing (when used orally). |
Legal Actions and Lawsuits
Some benzocaine lawsuits allege that manufacturers failed to warn customers about life-threatening risks, including methemoglobinemia. Other cases are related to the improper marketing of benzocaine teething products to infants younger than two years of age.
The Judicial Panel on Multidistrict Litigation has not bundled any of these product liability lawsuits into a multidistrict litigation (MDL). MDLs consolidate multiple similar cases into a single case to expedite the legal process.
If you or your child developed methemoglobinemia after taking benzocaine, you may be eligible to file a lawsuit. Drugwatch can connect you with an experienced product liability attorney to determine if you have a case.
Benzocaine Alternatives
Multiple topical anesthetics are safe and effective alternatives to benzocaine. If you have a benzocaine allergy, you may be able to use several other topical anesthetics on the market.
For example, lidocaine is a similar but stronger medicine than benzocaine. Consequently, it needs a lower concentration to be effective.
- Dibucaine
- Lidocaine
- Pramocaine
- Tetracaine
However, not all versions of these medications are appropriate for every part of your body. There are many versions of local anesthetics, including creams, rinses and injections. Read the label thoroughly before using any new medications.
For pain relief from infant teething, doctors generally recommend drug-free alternatives. These include massaging the gums and using a firm rubber teething ring.
To get relief from mouth sore pain, a saltwater and baking soda rinse may be effective. The ideal ratio is one teaspoon of baking soda and one teaspoon of salt for every four cups of water.
Calling this number connects you with a Drugwatch.com representative. We will direct you to one of our trusted legal partners for a free case review.
Drugwatch.com's trusted legal partners support the organization's mission to keep people safe from dangerous drugs and medical devices. For more information, visit our partners page.




