Spinal Cord Stimulator: Device Failures, Injuries and Safety Concerns
Spinal cord stimulators use electric impulses to help people control pain sensations. Despite widespread use, these devices carry documented failure risks that lawsuits against Medtronic, Abbott, Boston Scientific and Nevro say the manufacturers inadequately disclosed.
Our content is developed and backed by respected legal, medical and scientific experts. More than 30 contributors, including product liability attorneys and board-certified physicians, have reviewed our website to ensure it’s medically sound and legally accurate.
legal help when you need it most.
Drugwatch has provided people injured by harmful drugs and devices with reliable answers and experienced legal help since 2009. Brought to you by The Wilson Firm LLP, we've pursued justice for more than 20,000 families and secured $324 million in settlements and verdicts against negligent manufacturers.
More than 30 contributors, including mass tort attorneys and board-certified doctors, have reviewed our website and added their unique perspectives to ensure you get the most updated and highest quality information.
Drugwatch.com is AACI-certified as a trusted medical content website and is produced by lawyers, a patient advocate and award-winning journalists whose affiliations include the American Bar Association and the American Medical Writers Association.
About Drugwatch.com
- 17 Years of Advocacy
- $324 Million Recovered for Clients
- 20,000 Families Helped
- A+ BBB Rating
- 4.9 Stars from Google Reviews
Testimonials
I found Drugwatch to be very helpful with finding the right lawyers. We had the opportunity to share our story as well, so that more people can be aware of NEC. We are forever grateful for them.
- Last update: May 27, 2026
- Est. Read Time: 8 min read
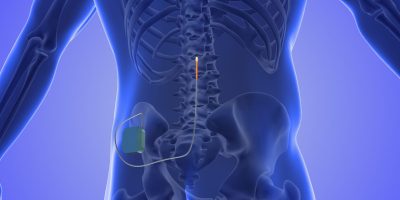
What Is a Spinal Cord Stimulator?
A spinal cord stimulator (SCS) is an implanted pain control system that uses mild electrical pulses to change how a person’s body feels pain. The system includes a small battery-powered pulse generator and thin wires called leads. A surgeon places the generator under the skin and positions the leads in the epidural space along the spine. The patient then uses a handheld remote to turn the system on and off and adjust settings.
Doctors usually consider a spinal cord stimulator for a person diagnosed with chronic pain that doesn’t respond to other treatments. Common uses include failed back surgery syndrome, complex regional pain syndrome (CRPS) and certain types of peripheral neuropathy. The stimulator doesn’t fix the source of pain. The device changes how nerves send pain signals so the person feels less pain and can move more easily. Many people still use other treatments, such as physical therapy or medicine, along with the device.
How Spinal Cord Stimulators Work
A spinal cord stimulator sends small electrical impulses through the leads to specific nerves in the spinal cord. It doesn’t eliminate pain, but the impulses block or change pain signals so the brain can tune them out. This allows people with chronic pain to focus on other activities, such as sleeping or working. A remote control lets the person control the spinal cord stimulator when they’re in pain.
Traditional spinal cord stimulators create a tingling feeling called paresthesia in the painful area. Many people describe this tingling as a gentle buzzing that replaces part of the pain. Newer sub-perception systems work at a level the person doesn’t feel. Those systems change pain signals without any tingling sensation. A doctor chooses the type of device and program based on the person’s diagnosis, pain pattern and lifestyle.
The Trial Period and Implant Procedure
A spinal cord stimulator procedure usually involves two stages. The first stage is a short trial that uses an external generator. A doctor places temporary leads in the epidural space through a needle. The leads connect to a small external device that the person wears for a determined trial period. During this time, the person tracks pain relief, daily activity and any problems. If the stimulator provides strong pain relief — reducing pain by at least half — the patient and doctor can move to a permanent implant.
The second stage is a surgery to implant the full system. A surgeon creates a small pocket under the skin in the abdomen or upper buttocks and places the generator there. The surgeon then threads permanent leads into the epidural space and connects them to the generator. Most people go home the same day or the next day.
Manufacturers and Device Types
Four primary companies produce spinal cord stimulators used in the United States. These companies include Medtronic, Abbott (formerly St. Jude Medical), Boston Scientific and Nevro. Each company offers several device lines with different features such as paresthesia-based programs, sub-perception (paresthesia-free) programs and rechargeable batteries.
These four manufacturers account for the majority of spinal cord stimulators that surgeons implant across the country. All four companies currently face active litigation related to spinal cord stimulator devices and complications.
Spinal Cord Stimulator Problems and Device Failures
About 50,000 spinal cord stimulator procedures happen each year in the United States. Despite this high use, devices from all four major manufacturers share a pattern of failures that has led to serious injuries and federal lawsuits.
- Continued severe baseline pain
- Difficulty with SCS system programming
- Ineffective stimulation
- Lead migration
- Nerve pain
- Painful revision surgeries due to lead migration and the SCS battery flipping
- Unpredictable shock sensations
In a survey published in Orthopedic Reviews, physicians reported that over 20% of patients who underwent SCS removal did so because of device complications. These complications included hardware-related problems such as device malfunction, lead migration or lead fracture, as well as biological problems such as pain or infection.
A person who lives with a spinal cord stimulator and new or worsening pain should talk with their doctor right away to check for device problems.
Lead Migration and Wire Displacement
Lead migration happens when the thin wires move away from their original spot in the epidural space. The person may lose pain relief in the original region and feel stimulation in a different part of the body. Because the device can’t control pain effectively, pain can feel more severe.
Device reports and medical studies identify lead migration as one of the most common spinal cord stimulator complications across brands. Doctors often need to reprogram the device or perform another surgery to move or replace the leads.
Electrical Shocks and Unintended Stimulation
People implanted with an SCS may experience tingling, buzzing or shock-like sensations. These shocks may happen during body position changes, exercise or other physical activity.
Multiple patients shared their experiences with spinal cord stimulator shocks and overheating. One patient reported to the FDA that the device overheated and shocked them even after they turned it off with the remote. The pain became debilitating, and they had the stimulator removed. The pain was so severe that they filed for disability.
Generator Failure and Battery Problems
The implanted generator functions as the power and control center for the spinal cord stimulator system. Generator failures can cause sudden loss of pain relief, unpredictable shocks or complete device shutdown. One patient reported that they received a faulty SCS generator and battery. It burned the paddle inside their spine and overstimulated them, causing scar tissue. They had multiple surgeries and product replacements.
“I now have additional complications and am losing more functions of my legs and cognitive functions, headaches and pain. The scars and lesions and complications have left me disabled and with forearm crutches,” the patient said.
Battery problems create another major source of frustration. Some people experience premature battery depletion that forces more frequent charging or early replacement surgery. Others face charging errors, overheating concerns or communication failures between the charger and implant. Multiple manufacturers have issued recalls or Urgent Medical Device Advisories for generators and batteries over the past several years, which shows a pattern of device and software defects across brands.
“SCS problems include “ineffective stimulation, nerve pain, unpredictable shocking sensations, lead migration, difficulty with programming of the SCS system, painful revision surgeries due to lead migration and the SCS battery flipping and a return of severe baseline pain.”
Serious Injuries From Spinal Cord Stimulator Failures
Spinal cord stimulator failures have led to serious injuries, including nerve damage, paralysis and infections.
Nerve Damage and Paralysis
Nerve damage and paralysis from a spinal cord stimulator are serious but less common complications. In rare cases, leads can injure spinal cord tissue and cause permanent neurological harm. Researchers have reported that SCS may cause changes in bladder and bowel control.
The needle, leads or paddles of SCS devices may cause trauma to the nervous system and affect the motor, sensory or autonomic nervous system, which may affect walking, balance and daily tasks.
Some reports describe weakness in both legs, known as paraparesis, after a trial or full implant.
Infections and Spinal Abscess
Every spinal cord stimulator implant carries a risk of infection. Infections associated with SCS are very serious because of the location of the device. Most people receive antibiotics before implantation. The implantable neurostimulator or receiver pocket are typically the most common sites of infection. In rare cases, these infections can occur years after implantation.
Signs of infection include redness, warmth, drainage or tenderness near the battery site.
Mild infections sometimes respond to antibiotics alone. More serious infections usually require device removal to fully clear the bacteria. In severe cases, the infection spreads deeper and forms an abscess near the spinal cord. A spinal cord abscess is a medical emergency and requires rapid surgery to remove the device and drain the infected area.
Permanent Restrictions After Implant
After an SCS implant, doctors often advise patients to avoid heavy lifting and twisting because this may displace the wires and interfere with device performance.
Because strong magnetic fields may disrupt the device, you may have issues getting an MRI. Newer spinal cord stimulators are supposed to be MRI-compatible, but you should speak with your doctor to be on the safe side. This is also an issue with all devices that emit strong electrical or magnetic fields.
If you plan to travel, make sure you carry documentation for your spinal cord stimulator and avoid metal detectors or security scanners that may interfere with the device.
Warning Signs Your Device May Be Failing
- Device no longer responding to programming
- Fever or swelling at the implant site
- Lack of pain relief
- New pain
- Sudden jolts or other electric sensations
If you have any of these issues or signs, contact your doctor or seek emergency medical attention right away.
FDA Recalls, Safety Alerts and Regulatory Oversight
Medtronic, Abbott and Boston Scientific have each faced FDA recalls or released safety communications related to lead integrity, generator malfunction or software errors.
- Medtronic (December 2025)
- Class 2 Device Recall for Vanta Clinician Programmer Application. The app didn’t function correctly and could prolong or postpone surgery. About 7,123 devices were affected.
- Abbott (October 2025)
- Class 2 Device Recall for Abbott Eterna. The devices may experience a loss of communication with the Clinician Programmer (CP) and/or the Patient Controller (PC). About 20,715 devices were affected.
- Boston Scientific (July 2024)
- Class 2 Device Recall for WaveWriter Alpha Spinal Cord Stimulation. The generator may reset during charging and lead to transient loss of stimulation and undesired sensations when therapy turns off and then back on. Patients may request surgical intervention for replacement or revision. About 77,674 devices were affected.
- Abbott (July 2023)
- Class I Recall for Proclaim and Infinity IPG. Devices couldn’t switch out of MRI mode. There were 73 reported injuries. Using the affected device may require surgery to remove it. About 155,028 devices were affected.
Nevro hasn’t had any recent recalls, but the FDA continues to receive reports of problems with the company’s devices.
Adverse Event Reports
The FDA has received tens of thousands of adverse event reports connected to spinal cord stimulators. According to an Associated Press investigation, the FDA received more than 80,000 injury reports from 2008 to 2018 alone.
Reported injuries include lead migration, electrical injury (shocks or burns), infection (sometimes requiring device removal), nerve damage, device failure requiring removal and others.
How These Devices Received FDA Clearance
The FDA approved many of the original spinal cord stimulators through the premarket approval program (PMA). This is the FDA’s most stringent medical device approval process and requires sufficient evidence that the device is safe and effective.
However, the FDA cleared some of these devices, parts or kits through the 510(k) premarket clearance process. The 510(k) requires less evidence and testing than a PMA approval before a product hits the market.
Spinal Cord Stimulator Lawsuits and Legal Action
People injured by spinal cord stimulators manufactured by Medtronic, Abbott, Boston Scientific and Nevro have filed lawsuits against the companies. These cases claim design defects in leads and generators, failure to warn patients about known malfunction risks and inadequate testing before market release.
People who used an SCS and experienced lead migration, device infection, nerve damage or device removal may qualify to file a lawsuit. For full eligibility criteria, multidistrict litigation (MDL) status and how to file, read more about spinal cord stimulator lawsuits.
Frequently Asked Questions About Spinal Cord Stimulators
Calling this number connects you with a Drugwatch.com representative. We will direct you to one of our trusted legal partners for a free case review.
Drugwatch.com's trusted legal partners support the organization's mission to keep people safe from dangerous drugs and medical devices. For more information, visit our partners page.