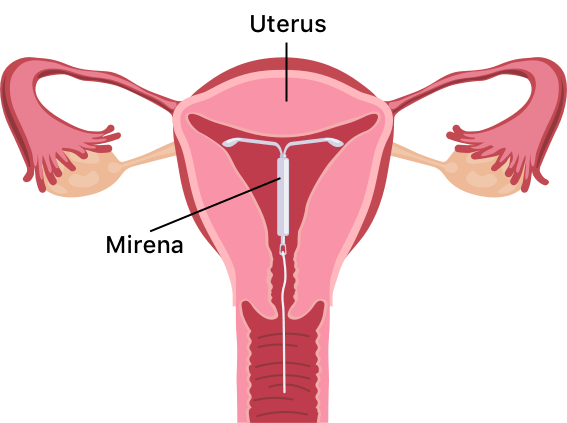
Mirena
Mirena birth control is a T-shaped intrauterine device (IUD) from pharmaceutical manufacturer Bayer. The IUD releases levonorgestrel, a hormone that thickens the mucus in the cervix to stop egg fertilization and partially suppresses ovulation.
Our content is developed and backed by respected legal, medical and scientific experts. More than 30 contributors, including product liability attorneys and board-certified physicians, have reviewed our website to ensure it’s medically sound and legally accurate.
legal help when you need it most.
Drugwatch has provided people injured by harmful drugs and devices with reliable answers and experienced legal help since 2009. Brought to you by The Wilson Firm LLP, we've pursued justice for more than 20,000 families and secured $324 million in settlements and verdicts against negligent manufacturers.
More than 30 contributors, including mass tort attorneys and board-certified doctors, have reviewed our website and added their unique perspectives to ensure you get the most updated and highest quality information.
Drugwatch.com is AACI-certified as a trusted medical content website and is produced by lawyers, a patient advocate and award-winning journalists whose affiliations include the American Bar Association and the American Medical Writers Association.
About Drugwatch.com
- 17 Years of Advocacy
- $324 Million Recovered for Clients
- 20,000 Families Helped
- A+ BBB Rating
- 4.9 Stars from Google Reviews
Testimonials
I found Drugwatch to be very helpful with finding the right lawyers. We had the opportunity to share our story as well, so that more people can be aware of NEC. We are forever grateful for them.
- Medically reviewed by Jessica D. Hess, Ph.D.
- Last update: May 19, 2026
- Est. Read Time: 10 min read
What Is Mirena?
Mirena birth control is a T-shaped intrauterine device, or IUD, that releases the hormone levonorgestrel to thicken the mucus within the cervix, preventing the fertilization of eggs. The hormone also thins the endometrium (the lining of the uterus), which can partially suppress ovulation.
The Bayer-manufactured Mirena device is the No. 1 prescribed IUD in the US. A health care professional must place this soft plastic device in (and remove it from) your uterus. You will need to check its thin strings on the bottom yourself once inserted to ensure it is still in place.
Mirena contains a reservoir of levonorgestrel hormone. It lasts up to eight years for pregnancy prevention and eliminates the responsibility of taking a daily pill.
Levonorgestrel vs. Copper IUD
Long-acting, reversible contraception via IUD comes in two options: the hormonal levonorgestrel IUD and the copper IUD. Both are very effective.
The copper IUD has an efficacy rate of 99.2%, so out of every 1,000 people, eight may have an unintended pregnancy. The copper IUD does not contain hormones. It works because the properties of copper affect egg and sperm motility and survival.
A levonorgestrel IUD has a slightly higher efficacy rate of 99.8%, which means only two people out of every 1,000 may have an unintended pregnancy.
How Does Mirena Work?
Mirena birth control releases levonorgestrel, a type of synthetic progestin often used in birth control pills. The device slowly releases a continuous low dose of the hormone into your uterus and bloodstream.
Levonorgestrel thins the lining of the uterus and thickens the mucus in the cervix, making it difficult for sperm to move into and survive in the uterus. The thinning of the uterine lining also makes it less likely for an egg to attach to the uterus.
According to Bayer, Mirena is more than 99% effective at preventing pregnancy. The U.S. Food and Drug Administration (FDA) has approved this drug to prevent pregnancies for up to eight years.
Mirena Insertion and Removal
Your doctor can insert the Mirena device during a regular doctor’s visit. Mirena insertion takes about five minutes.
You should wait at least six weeks to start using Mirena if you have just given birth, had a miscarriage or had an abortion.
A health care provider will need to remove the Mirena IUD after eight years when it is no longer effective. If the device moves out of place, your provider can insert a new one.
Mirena has no long-term effect on your ability to conceive. Eight out of 10 women who have had their Mirena IUD removed can get pregnant within a year.
Pros and Cons of Mirena IUD
Like any method of birth control, Mirena has pros and cons. On the positive side, it’s highly effective and begins working immediately.
“I will say there are times when Mirena is needed, and it is often a better choice than surgery or full-dose hormonal birth control.”
It’s so tiny you cannot feel it, nor can your partner. Mirena is also low-maintenance, as you don’t have to remember to do anything for it to function. And, in most cases, it is entirely and almost instantly reversible if you decide to get pregnant.
For some, one con is that a physician or midwife must insert it. You may experience some cramping immediately after insertion. Additionally, the IUD does not protect against sexually transmitted diseases.
Benefits of Mirena
Mirena was the first hormone-releasing IUD to gain FDA approval to treat heavy periods for up to five years in women who choose an IUD for birth control.
Mirena thins the lining of the uterus, which can lessen heavy menstrual bleeding. In one clinical trial, Mirena reduced menstrual blood loss by more than 50% after six months in nine out of 10 women. Mirena can also decrease menstrual pain and pain related to endometriosis.
A 2022 clinical study of 80 participants studied the effects of the Mirena IUD on endometrial thickness and the ability to cure patients with perimenopausal abnormal uterine bleeding. The endometrial thickness and menstrual volume scores following three months of treatment were remarkably lower than those before treatment and those of the control group.
- Mirena is more than 99% effective.
- You don’t have to worry about remembering to take a pill.
- It lasts up to eight years for contraception, according to the FDA.
- Most insurance plans cover Mirena.
- It is long-term and reversible birth control.
- Women can still breastfeed while using Mirena.
- It uses a lower dose of hormones than some other birth control methods.
- It reduces or stops menstrual flow in women who don’t want a period or suffer from excessive bleeding.
Experts also recommend IUDs such as Mirena as a birth control option for teens and younger adults because they are effective, need no daily care and last for years.
Disadvantages of Mirena
As with any birth control method, you should weigh the risks and benefits before using Mirena. One of the disadvantages of Mirena is that it may cause ovarian cysts.
Becoming pregnant while using Mirena can be life-threatening for the mother and baby under certain circumstances.
- It may cause ovarian cysts.
- Pregnancy with Mirena can be life-threatening.
- IUDs can become infected during insertion.
- Device insertion can cause perforation.
- It doesn’t protect against STDs.
- Device expulsion can occur, leaving the woman at risk of pregnancy.
IUDs may lead to severe infection or pelvic inflammatory disease. In addition, perforation of the uterine wall or cervix can occur.
For more information, speak to your doctor about the risks of using Mirena.
Mirena Side Effects
The most common side effects of Mirena include pain, ovarian cysts, menstrual changes and acne.
- Abdominal or pelvic pain
- Acne
- Allergic reactions such as rash, hives, difficulty breathing or facial swelling
- Breast tenderness
- Changes in bleeding, such as spotting or heavier bleeding within the first 3-6 months
- Depression or changes in mood
- Hair loss or unwanted hair growth
- Headache or migraine
- Missed menstrual periods
- Nausea
- Ovarian cysts that typically resolve within 2-3 months
- Pain, bleeding or dizziness during and after placement
- Vaginal discharge
- Vulvovaginitis (genital inflammation)
- Weight gain
Some side effects can be serious. If you experience any signs of an allergic reaction, contact your care provider immediately.
Managing Side Effects of Mirena
Over-the-counter nonsteroidal anti-inflammatory drugs (NSAIDs) like ibuprofen, naproxen or aspirin can usually help with IUD cramps. Additionally, period products such as panty liners or pads may help reduce the incidence of blood seeping into your clothes.
If you have cramping or bleeding that doesn’t improve within a few months, talk with your doctor, as they may need to check the placement of your IUD.
Serious Side Effects of Mirena
The more severe side effects that affect Mirena users, such as pregnancy complications, device migration and infection, can be serious and even life-threatening.
- Breast cancer
- Device expulsion, migration or perforation
- Ectopic pregnancy
- Miscarriage
- Pelvic inflammation
- Pseudotumor cerebri (PTC), also called idiopathic intracranial hypertension (IIH)
- Sepsis
Patients have filed thousands of Mirena lawsuits against Bayer for perforation, ectopic pregnancy and PTC, which is increased pressure in the skull.
Ectopic Pregnancy
According to Mirena’s Frequently Asked Questions, roughly one-half of all pregnancies that occur with Mirena are likely to be ectopic. An ectopic pregnancy occurs when a fertilized egg begins to develop into a fetus outside of the uterus, typically in the fallopian tubes. Without treatment, this condition has a high mortality rate for the pregnant person. However, even with treatment, the fetus is likely to die.
People who have previously experienced an ectopic pregnancy, pelvic infection or tubal surgery have an increased risk of ectopic pregnancy while using Mirena.
Perforation and Migration
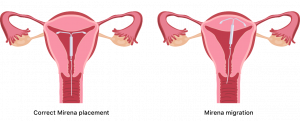
Mirena migration is when the IUD shifts to another part of the body or leaves the uterus.
Uterine perforation, which is when the device erodes through your uterus, occurs most often during insertion. However, it may go undetected at first. Perforation risk is higher in those who have recently given birth or are breastfeeding.
Delayed removal of Mirena in cases of perforation may result in the device migrating outside of the uterine cavity.
Expulsion
Complete or partial expulsion of Mirena may occur, rendering it ineffective as a contraceptive. Expulsion may cause symptoms of bleeding or pain, but it may also be asymptomatic and go unnoticed.
The cause of expulsion is usually unknown, but people who have never been pregnant or have heavy periods are more likely to experience this side effect.
Sepsis
Sepsis, which is a severe response to an infection, can cause organ damage if left untreated. The Sepsis Alliance T.I.M.E. guidelines teach people how to detect sepsis. High temperature, symptoms of infection, altered mental state and extreme illness are potential symptoms of sepsis. Contact your care provider immediately if you experience any symptoms.
Spontaneous Abortion of Intrauterine Pregnancy
Patients who have become pregnant with an IUD in place may experience a septic abortion, in which septicemia (blood poisoning), septic shock and death may occur.
If you become pregnant, contact your provider right away to have Mirena removed promptly. Leaving Mirena in place may increase the risk of spontaneous abortion and preterm labor.
Never remove Mirena on your own. Aside from the risk of the device getting stuck, probing the uterus by shifting the device and removing Mirena incorrectly may also result in spontaneous abortion. Only allow a professional to remove your device.
Pelvic Infection
Pelvic or lower abdominal pain, foul-smelling vaginal discharge, bleeding, fever and genital sores or lesions are potential signs of infection. Your doctor will likely remove Mirena if the pelvic infection is severe or does not respond to treatment.
Pelvic Inflammatory Disease (PID)
There is an increased risk of PID in IUD users, most likely due to organisms being introduced to the uterus during insertion. In clinical trials, 3.5% of Mirena users reported upper genital infections, which occurred more frequently in the first year.
Pseudotumor Cerebri (PTC)
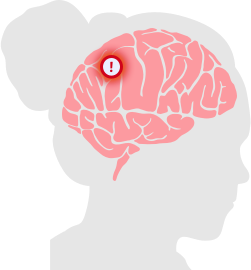
Intracranial hypertension is also known as a “false brain tumor.”
Hormonal IUDs like Mirena put users at an increased risk for pseudotumor cerebri (PTC), also known as a false brain tumor or idiopathic intracranial hypertension (IIH). PTC occurs when there is increased pressure between the skull and brain that isn’t caused by a mass or lesion. This high pressure in the skull can be due to the buildup or poor absorption of cerebrospinal fluid. PTC can cause vision loss from swelling within the eyes, debilitating headaches and reduced quality of life.
You should report symptoms like headaches, vision issues and ringing in the ears to your doctor immediately.
Mirena FDA Warnings
The most recent FDA warnings about Mirena are in Mirena’s 2008 labeling updates. These warnings cover several of its severe side effects. In addition to ectopic pregnancy, sepsis, PID, perforation and expulsion, the FDA warnings mention the following adverse events.
- Breast cancer
- In Mirena’s postmarketing experience, there were multiple reports of breast cancer. Because breast cancer is hormone-sensitive, people with a history or suspicion of this cancer should not take hormonal IUDs.
- Embedment
- Mirena IUDs may become embedded in the uterine wall, decreasing their contraceptive effects. Sometimes, embedded IUDs require surgical removal.
- Intrauterine pregnancy
- If a non-ectopic pregnancy occurs with a Mirena IUD in place, there are still several risks, including sepsis, miscarriage and premature labor.
- Irregular bleeding or amenorrhea
- Mirena IUDs can affect regular bleeding patterns, resulting in irregular bleeding, spotting, amenorrhea and similar concerns.
- Ovarian cysts
- Mirena IUDs may increase the risk of ovarian cysts in some patients. Usually, they will disappear on their own within a few months.
- Risks of mortality
- While IUDs are a safe form of birth control overall, they still have a small risk of death. It is important to fully understand this risk before selecting which type of birth control to use.
Patients must consider these potential risks and their own medical history to evaluate whether the Mirena IUD is the best option for them.
Who Should Not Use Mirena?
Women who are pregnant or suspect they might be pregnant should not use Mirena. Several gynecological conditions make using Mirena ill-advised or dangerous. These include a history of pelvic inflammatory disease, postpartum endometriosis, a recent infected abortion, abnormal pap smears, genital bleeding or infections and uterine abnormalities, such as fibroids.
Individuals with a history of liver disease, liver tumors, breast cancer or other hormone-sensitive cancers should avoid using Mirena.
If you have known hypersensitivities to the IUD or any of its components, avoid Mirena and consider other birth control options.
Mirena Interactions
When you take two or more medicines at the same time, the effects of one medicine can interact with the others. It’s important, therefore, to tell your doctor what medications you are taking.
Certain drugs and herbal supplements can make Mirena less effective. Examples of these substances include some blood thinners, anti-anxiety drugs and antiseizure medications. Antiretroviral drugs, antibiotics and St. John’s wort may also interact with Mirena.
How Much Does Mirena Cost?
Some insurance plans cover the costs of Mirena, while people without insurance could pay over $1,000. Under the Affordable Care Act, plans in the health insurance marketplace must cover some type of contraception.
Bayer has indicated that its surveys show 95% of women received insurance coverage for an IUD like Mirena, with little to no out-of-pocket costs.
Calling this number connects you with a Drugwatch.com representative. We will direct you to one of our trusted legal partners for a free case review.
Drugwatch.com's trusted legal partners support the organization's mission to keep people safe from dangerous drugs and medical devices. For more information, visit our partners page.




