Morcellator Advocate: Dr. Amy Reed
Dr. Amy Reed, a Boston-area mother of six, died in May 2017 after a battle with cancer, a disease that progressed rapidly after she underwent a procedure with a device called a power morcellator. Before her death, Reed and her husband became activists against the device, influencing Congressional legislation, sparking lawsuits and prompting the withdrawal of the device.
Our content is developed and backed by respected legal, medical and scientific experts. More than 30 contributors, including product liability attorneys and board-certified physicians, have reviewed our website to ensure it’s medically sound and legally accurate.
legal help when you need it most.
Drugwatch has provided people injured by harmful drugs and devices with reliable answers and experienced legal help since 2009. Brought to you by The Wilson Firm LLP, we've pursued justice for more than 20,000 families and secured $324 million in settlements and verdicts against negligent manufacturers.
More than 30 contributors, including mass tort attorneys and board-certified doctors, have reviewed our website and added their unique perspectives to ensure you get the most updated and highest quality information.
Drugwatch.com is AACI-certified as a trusted medical content website and is produced by lawyers, a patient advocate and award-winning journalists whose affiliations include the American Bar Association and the American Medical Writers Association.
About Drugwatch.com
- 17 Years of Advocacy
- $324 Million Recovered for Clients
- 20,000 Families Helped
- A+ BBB Rating
- 4.9 Stars from Google Reviews
Testimonials
I found Drugwatch to be very helpful with finding the right lawyers. We had the opportunity to share our story as well, so that more people can be aware of NEC. We are forever grateful for them.
- Last update: May 8, 2026
- Est. Read Time: 9 min read
Amy Reed was in her early 40s when she sought advice from gynecological oncologist Michael Muto for the pain and bleeding she had been experiencing since the birth of her youngest son, Ryan. That decision led her down an unexpected path that raised public awareness about a dangerous device — power morcellators — and ultimately led to her death.
Muto diagnosed Reed with a fibroid (a benign — or non-cancerous — tumor in the uterus), and referred her to his colleague, Karen Wang, a gynecological surgeon at Brigham and Women’s Hospital in Boston, Mass., where Reed herself was working at the time.
Although Reed was all for “traditional open-abdomen surgery,” Muto and Wang allegedly talked her out of it, opting instead for a less-invasive laparoscopic procedure using a power morcellator, she later told Boston Magazine.
Reed later learned the morcellation (or chopping up) of the fibroid stirred up hidden cancer cells and spread them throughout her abdomen.
When she underwent the procedure on October 17, 2013, she was told she would get her “back on [her] feet” quickly.
Wang phoned Reed about a week later with the news that the pathology report on her fibroid showed an unusually aggressive form of cancer, a type that doesn’t show up on preoperative tests, called leiomyosarcoma.
Muto also contacted Reed to inform her that she would need to undergo another surgery — this time to remove her ovaries. By March 2016, Reed had undergone four additional operations to remove new tumors.
At the time of receiving the news of her cancer, Reed and her husband, cardiothoracic surgeon Hooman Noorchashm, said this was also the first time they had heard of a fibroid being morcellated rather than being removed whole.
Noorchashm said that he just assumed any mass would be treated the same way. It’s best, he said, to take them out in one piece.
“The issue with the power morcellator is not that it has a mechanical flaw, but that it’s designed based upon an incorrect assumption and surgical protocol.”
While Reed and Noorchashm acknowledged that the power morcellator did not cause Reed’s cancer, they pointed out that the act of morcellation very likely spread the disease.
“If the sarcoma had been taken out whole, [Reed’s] five-year survival rate would have been 50 percent. With the cancer morcellated, it was between zero to 20 percent.” Noorchashm told Boston Magazine.
Who Was Dr. Reed?
Noorchashm and Reed met at Penn State in 1995, where they were both completing doctorate degrees. Prior to attending Penn for undergraduate and graduate coursework, Reed attended Villa Joseph Marie, a Catholic school, in Bucks County, Pennsylvania, where she was valedictorian. At Penn State, Reed pledged Phi Beta Kappa, she was Golden Key and she made the dean’s list.
After acquiring a doctorate in immunology, Reed applied to medical school and specialized in critical care and anesthesiology, a dual-board certification.

But most of all, having grown up in a home with eight siblings, Reed always wanted a big family. Reed and Noorchashm married in 2001 and had six kids in all, with three of those kids already born by the time Reed graduated from medical school and two more following by the time the couple finished their medical residencies in 2011. Ryan was the last of the brood leading up to Reed’s surgery.
Reed’s career included a teaching post alongside her husband at Harvard Medical School, a book titled “Naughty Children,” and treatment of Boston Marathon bombing victims and wounded bomber Dzhokhar Tsarnaev.
Husband and Wife Battle Against the System
Noorchashm told Drugwatch his wife was the first person to submit an adverse event report to the U.S. Food and Drug Administration (FDA) about power morcellators. That, along with an influx of emails sent by Noorchashm to physicians, government regulators, legislators, insurers, hospital administrators, reporters and anyone else who might lend an ear to their cause, resulted in several changes to the use of power morcellators in the gynecological community.
According to a 2022 research study, laparoscopic power morcellator use decreased by 9.5% for each quarter elapsed after the FDA warning period.
Philly.com said “[Reed] was initially uncomfortable being an activist, but came to feel she and her husband were uniquely equipped to challenge entrenched medical interests, given their training, savvy and sense of duty.”
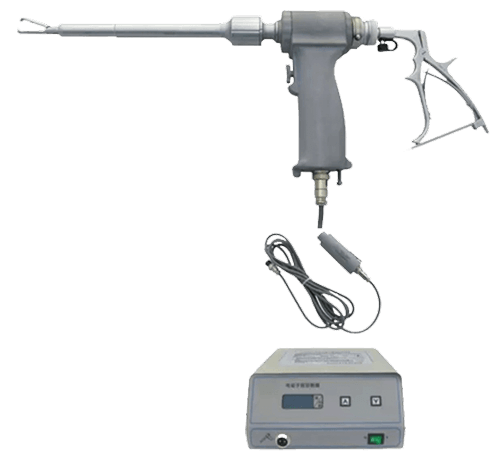
Following Reed’s cancer diagnosis, a friend from medical school, Michael Paasche-Orlow reviewed scientific literature and collected data. His work led to the FDA’s most recent risk estimate of the morcellator risk. The FDA says 1 in 350 women could potentially be harmed by the spread of uterine cancer after the use of a power morcellator in laparoscopic surgical procedures. The previous risk estimate was 1 in 10,000.
After Reed’s case became public, more women stepped forward about their own cases. That resulted in hospitals limiting the use of power morcellators, including the hospital where Reed’s procedure was performed. In 2014, the FDA issued a safety communication discouraging the use of power morcellators for fibroid surgery.
In July of the same year, Johnson & Johnson withdrew its morcellators from the market. Some insurers, including giant Blue Cross Blue Shield, refused to pay for hysterectomies performed with power morcellators . And in November, the FDA, for the first time, exercised its “immediately in effect” authority (bypassing public comment) requiring a black box warning, its most serious type of warning, to be included on all morcellators. The warning advised patients and health care providers that the devices are contraindicated for removal of fibroids or uterine tissue.
Noorchashm faced backlash from his colleagues. His employer, Brigham and Women’s Hospital, told other workers to have no contact with him.
When they returned to the hospital for a follow-up operation for Reed, Noorchashm had to obtain a restraining order against the facility because they were searched and a security guard accompanied Noorchashm.
But Noorchashm and Reed continued in their fight, eventually gaining attention and support from U.S. House Reps. Mike Fitzpatrick and Louise Slaughter. After 12 congressmen prompted the Government Accountability Office (GAO) to investigate potential negligence by the FDA in the clearance and surveillance of power morcellators, Fitzpatrick and Slaughter also sponsored a bill proposed by Noorchashm and Reed to address the current issues with the FDA adverse reporting system.
Medical Device Guardians Act
A bill called the “Medical Device Guardians Act of 2016” was introduced in the House of Representatives on June 8, 2016, and is currently pending before the House Committee on Energy and Commerce.
This bill, proposed by Noorchashm and his late wife and sponsored by Fitzpatrick and Slaughter, would require doctors and hospitals to report adverse events associated with medical devices but it would protect them from potential liability exposed by their reports.
Noorchashm and the bill’s sponsors assert that these individuals are the “the best people to report that back to the FDA.” But he also pointed out, as a medical practitioner himself, that he understands the fear of liability as he himself used to regard medical malpractice and personal injury lawyers in a bad light. But he now understands that they can help when patients are harmed.
As Noorchashm and Reed previously reported to one media source, “It is the fear of liability and the FDA’s lax enforcement of ‘self-reporting’ requirements that has created a ‘culture of complacency’ in the medical device space.”
“It is the fear of liability and the FDA’s lax enforcement of ‘self-reporting’ requirements that has created a ‘culture of complacency’ in the medical device space.”
They continued, “Legislation that would protect individual practitioners from liability when reporting adverse outcomes associated with the use of medical devices, and prosecuting failures to ‘self-report’ would reveal problems very quickly.”
Sponsors Fitzpatrick and Slaughter told The Wall Street Journal that the responsibility to report the problem should not have fallen on the patients, that it should not be their duty to alert the FDA. In a letter introducing the bill, the representatives stated,” ‘Everyone in the chain of care’ should be responsible for reporting adverse events to FDA.”
Overriding Medical Ethics
Noorchashm said that “the crux of [Reed’s] fight came down to this idea that you cannot assert avoidable mortality risks in medicine.” In the instance of power morcellators, Noorchashm said even at less than 1 percent affected, these women’s lives are jeopardized and it’s considered collateral damage. But he pointed out that with approximately 600,000 operations performed each year, thousands of women are now put in harm’s way and that is not a “negligible number.”
Even in 2017, “medicine is still operating under a “defunct and incorrect thinking” of utilitarianism basically justifying the sacrifice of a minority number of lives since the majority are benefiting,” says Noorchashm, adding “that’s not OK… we don’t do that… it overrides medical ethics.”
Noorchashm’s Message: “Let the War Begin”
On his blog, Noorchashm recently said, “Back in 2013… I could have never imagined writing an article like this one. But this all changed on May 24, 2017 when my beloved wife lost her life, unnecessarily, following a medical complication.”
Noorchashm’s message included an invitation: “The complacency and protectionism exhibited by highly decorated leaders in gynecology and corporate medical leaders has left me no choice but to write this article and to invite every plaintiffs’ attorney in America to engage what will be a winnable fight to bring sanity to gynecology — and to recover the damage that is being done to many women and families.”
“With Amy’s passing, the word morcellation will become synonymous with medical malpractice and negligence for posterity… Let the war begin.”
In his interview with DrugWatch, Noorchashm explained that gynecologists, as “poorly trained surgeons” cannot violate a core principal of surgery in order to perform small incision surgery – being that “gynecologists fundamentally violate [the] principal” that a tumor can never be assumed to be benign.
Noorchashm noted that even with power morcellators being removed from the practice, gynecologists will find other methods for morcellating these fibroid tumors, using a standard scalpel procedure. Noorchashm says it’s imperative that the medical community starts “using a different language” — not that power morcellators are the only evil, but that any kind of morcellation can impose the same risk of harm to women, “exposing that woman to the chance of the cancer being spread and upstaged.”
A Legacy Left Behind
While Reed has left behind her husband Noorchashm, six children (including four sons and two daughters), her parents and seven siblings, Noorchashm believes she has also left behind a battle for women that speaks for itself. Noorchashm says that because of his wife, her experience with a power morcellator and a public health campaign started by the couple in 2013, several changes have transpired impacting health care for women.
- FDA actions to restrain power morcellators in the marketplace
- Johnson & Johnson (the largest producer of power morcellators) withdrawing its device from the market
- Most large medical centers nationwide no longer using power morcellators
- Settlement of hundreds of lawsuits against J&J
- Ongoing lawsuits pending against gynecologists and remaining major power morcellator manufacturer STORZ
“Amy’s fight speaks for itself,” Noorchashm said. “I believe our kids will learn from it and be proud of her.”
Calling this number connects you with a Drugwatch.com representative. We will direct you to one of our trusted legal partners for a free case review.
Drugwatch.com's trusted legal partners support the organization's mission to keep people safe from dangerous drugs and medical devices. For more information, visit our partners page.


