Humira Lawsuits
People who used Humira to treat symptoms of rheumatoid arthritis, Crohn’s disease, psoriatic arthritis and other inflammatory conditions have sued the drug’s maker over allegations that it failed to adequately warn of the risks, namely invasive fungal infections, nerve damage and cancer.
Our content is developed and backed by respected legal, medical and scientific experts. More than 30 contributors, including product liability attorneys and board-certified physicians, have reviewed our website to ensure it’s medically sound and legally accurate.
legal help when you need it most.
Drugwatch has provided people injured by harmful drugs and devices with reliable answers and experienced legal help since 2009. Brought to you by The Wilson Firm LLP, we've pursued justice for more than 20,000 families and secured $324 million in settlements and verdicts against negligent manufacturers.
More than 30 contributors, including mass tort attorneys and board-certified doctors, have reviewed our website and added their unique perspectives to ensure you get the most updated and highest quality information.
Drugwatch.com is AACI-certified as a trusted medical content website and is produced by lawyers, a patient advocate and award-winning journalists whose affiliations include the American Bar Association and the American Medical Writers Association.
About Drugwatch.com
- 17 Years of Advocacy
- $324 Million Recovered for Clients
- 20,000 Families Helped
- A+ BBB Rating
- 4.9 Stars from Google Reviews
Testimonials
I found Drugwatch to be very helpful with finding the right lawyers. We had the opportunity to share our story as well, so that more people can be aware of NEC. We are forever grateful for them.
- Medically reviewed by Tess Schulman, Ph.D.
- Last update: May 8, 2026
- Est. Read Time: 8 min read
People who say they or their loved ones experienced life-altering effects from Humira sued its manufacturer, Abbott Laboratories (now spun off as AbbVie Inc.), claiming negligence.
- Invasive fungal infections, including pulmonary or disseminated histoplasmosis
- Optic neuritis
- Peripheral neuropathy
- Leukemia
- Hepatosplenic T-cell lymphoma
Plaintiffs argued the damage the drug caused them could have been prevented or at least diagnosed sooner had Abbott properly warned of the risks.
As of May 2026, most attorneys were no longer accepting these types of Humira cases.
Fungal Infection Lawsuits
In September 2008, the FDA warned Abbott that its blockbuster medicine Humira caused serious, life-threatening fungal infections that were initially unrecognized by doctors.
“This has resulted in delays in appropriate treatment, sometimes resulting in death,” the agency said.
The FDA ordered Abbott to provide new information about the risk of unrecognized, drug-induced invasive fungal infections — such as pulmonary and disseminated histoplasmosis, coccidioidomycosis and blastomycosis — in the black box warning and warnings sections of the drugs’ prescribing information and the Medication Guide for patients. The agency also required Abbott to educate prescribing doctors about this risk.
However, the drug company allegedly waited almost two years before sending letters to doctors about the risk. Records show Abbott didn’t update its black box warning until November 2009 and didn’t send a warning letter to doctors about the risk until May 17, 2010.
As a result, people filed lawsuits that accused the company of failing to adequately warn about the drug’s unrecognized, drug-induced fungal infections even after receiving reports of patient deaths.
First Fungal Infection Trial
Like thousands of others, Delores Tietz used Humira as prescribed for her rheumatoid arthritis. She injected the medicine for nearly seven months, beginning in October 2009. However, in May 2010, Tietz began experiencing chest pain and fevers, according to court documents.
At first, Tietz illness went undiagnosed, and doctors told her family she was unlikely to survive. After 21 days in the hospital, doctors determined Tietz suffered from Humira-induced disseminated histoplasmosis, a severe, life-threatening fungal infection.
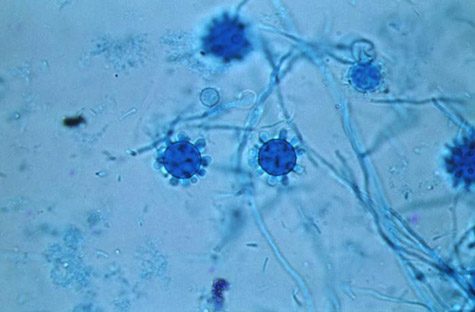
Tietz would go on to survive for roughly three years, and in 2012, she sued the drug’s manufacturer for allegedly failing to warn her or her doctor of the risk of histoplasmosis infection. Attorneys for Tietz claim that despite an FDA alert in 2008, the company failed to send warning letters directly to physicians until May 17, 2010 — 10 days after Tietz entered the hospital with an infection.
When Tietz died in March 2013, her husband, Milton, took over the lawsuit. The case, Milton Tietz, et al. v. Abbott Laboratories, et al., went to trial in April 2013 in Illinois. Judge William J. Haddad oversaw the proceedings. Attorneys for Tietz said the case was the first Humira injury case to go to trial.
“Humira has dangerous side effects and Abbott was negligent regarding them,” an attorney for Tietz said in a press release. “Justice has been done.”
Following the verdict, AbbVie spokeswoman Adelle Infante said in a statement that the company planned to appeal.
“Humira has more than 15 years of clinical and safety data, with therapeutic risks well documented in the prescribing label,” the statement said.
AbbVie did not win its appeal. The court also found that despite its insistence, Abbott cannot use the FDA or its regulations to avoid its duty of ordinary care under state law. Judgment was entered against Abbott and the jury’s verdict against the company stands.
Histoplasmosis Case Filed in Tennessee
At age 69, Frederick Delano sued Abbott Laboratories in Tennessee, claiming his near-fatal case of histoplasmosis might have been diagnosed sooner or even prevented had the drug company not “stalled” on issuing a warning that the drug could cause serious fungal infections.
As part of its September 2008 communications, the FDA ordered the manufacturer to warn patients and doctors of an increased risk of histoplasmosis in Humira patients. The agency said the majority of cases were from Histoplasma capsulatum-endemic areas in the Ohio and Mississippi River valleys.
Delano, who lived in that region, began using Humira as prescribed for his psoriatic arthritis in October 2008. Delano was then hospitalized and diagnosed with disseminated histoplasmosis.
“In 2008, Fred and his physicians got no warning whatsoever that the medication which he thought would help could, in fact, kill him — and nearly did,” Delano and his wife, Frances, alleged in their complaint.
In addition, the manufacturer waited until November 2009 — more than a year after the FDA’s notice — to update the drug’s black box warnings, and until May 17, 2010 to send a warning letter to doctors, according to the complaint.
Delano filed the lawsuit on June 12, 2011. Judge William G. Young dismissed the case on September 28, 2012.
Nerve Damage Claims
In 2011, Humira’s manufacturer faced at least two lawsuits involving allegations that the drug caused nerve damage in patients with Crohn’s disease.
Kara Mae Pletan filed her lawsuit in state court in Chicago. Pletan claims she used Humira to treat symptoms of her Crohn’s disease and that the therapy caused permanent nerve damage in her feet (peripheral neuropathy).
Less than a month after Pletan filed her lawsuit against Abbott, Jamie Bixby also sued the company. Like Pletan, Bixby claims she used Humira to treat symptoms of her Crohn’s disease. Her lawsuit filed in May 2011 in Chicago federal court alleges the medicine permanently damaged her optic nerves.
The doctor who treated Bixby in 2008 said her optic neuritis was “related to her use of Humira,’’ according to the lawsuit. Bixby argues that other competitor drugs in the same class carried a specific warning about the risks of optic nerve damage, but for years Humira did not.
The drug’s label was updated in March 2011 to include a warning that users may experience new or worsening symptoms of optic neuritis. The current label now warns that the medicine has also been associated with central nervous system demyelinating disease, including multiple sclerosis (MS), and peripheral demyelinating disease, including Guillain-Barré syndrome.
Cancer Cases
Others who sued Abbott Laboratories say the company knew cancer could affect Humira users but failed to warn them about the risk.
One Texas lawsuit for example, claims Abbott knew since 2005 that people who take the drug have a three- to five-fold potential risk of cancer but did nothing to warn patients directly until the FDA required it in 2009.
In that case, Gayathri Murthy sued Abbott over allegations that she developed lymphoma after taking Humira in a company-sponsored clinical trial beginning in early 2005.
According to the complaint, Murthy’s doctor warned her that the medication increased the risk of tuberculosis and said the other side effects were similar to the “side effects of aspirin.’’ Murthy, who has rheumatoid arthritis, continued to use the medicine until she was diagnosed with lymphoma in February 2006, the suit says.
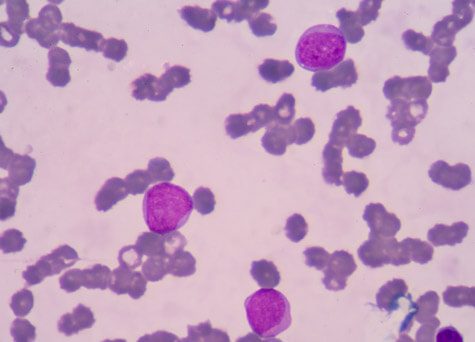
Another lawsuit filed in Texas claims the drug caused an 11-year-old boy’s leukemia. Bo Anderson used Humira as prescribed to treat his psoriasis, the suit says. After about 10 months, the boy’s parents noticed unusual bruising on his body. Doctors diagnosed the boy with T cell acute lymphoblastic leukemia in January 2009.
Bo’s diagnosis came more than seven months after the FDA said manufacturers like Abbott should warn physicians and patients about the risk of cancer, and nearly 10 months before the black box warning was finally issued regarding leukemia in children.
Today, the drug’s black box warning states: “Lymphoma and other malignancies, some fatal, have been reported in children and adolescent patients treated with TNF blockers including Humira.”
It also warns that cases of hepatosplenic T-cell lymphoma have occurred in adolescents and young adults with inflammatory bowel disease treated with TNF blockers, including Humira.
Kickbacks Lawsuit
In September 2018, the State of California filed suit against AbbVie, alleging the drugmaker gave “illegal kickbacks” to physicians to encourage them to prescribe Humira. According to a complaint filed by the California Insurance Commissioner, the pharmaceutical company provided physicians with cash, meals, gifts, trips, patient referrals and other incentives to motivate them to prescribe the drug.
This complaint also accused the company of using registered nurse “Ambassadors” who had one goal in mind: to “keep patients on a dangerous drug at any cost.”
“Ambassadors provide unbalanced information: trained to tout the good while at the same time instructed on methods to avoid directly answering patients’ questions on the bad, even those pertaining to Humira’s serious and important side effects,” the suit alleged.
These sort of “white coat marketing” techniques, as they’re sometimes called, are heavily frowned upon because they prey on the exceptional trust level most patients have in their clinicians.
Many of the claims in the suit were based on the allegations of a whistleblower, Lazaro Suarez, who had previously worked as a Humira nurse ambassador for AbbVie in Florida. According to the complaint, Suarez said he was taught to deflect patients’ questions about potential side effects of Humira and to focus instead on the fact that he could help patients receive their Humira for $5 or less.
The suit between the State of California and AbbVie was settled in 2020. Requirements of the settlement included reforms of AbbVie’s marketing practices, and the company was ordered to pay $24 million to the State of California and Suarez.
Calling this number connects you with a Drugwatch.com representative. We will direct you to one of our trusted legal partners for a free case review.
Drugwatch.com's trusted legal partners support the organization's mission to keep people safe from dangerous drugs and medical devices. For more information, visit our partners page.




