Blood Thinners
Blood thinners reduce the risk of blood clots. Doctors prescribe them for conditions like atrial fibrillation, heart disease and deep vein thrombosis. However, blood thinners can cause severe side effects, including excessive and internal bleeding, which can be life-threatening.

What Are Blood Thinners?
Blood thinners reduce the clotting factor of blood. They are typically used for people at higher risk of heart attack, stroke or blood clots.
- Aspirin
- Brilinta (ticagrelor)
- Coumadin (warfarin)
- Effient (prasugrel)
- Eliquis (apixaban)
- Plavix (clopidogrel)
- Pradaxa (dabigatran)
- Savaysa (edoxaban)
- Xarelto (rivaroxaban)
“Common side effects of blood thinners, like bruising and increased bleeding, are significant concerns,” Sazan Sylejmani, owner and pharmacy manager at Westmont Pharmacy in Westmont, Ill., told Drugwatch. “Patients can minimize these risks by using soft toothbrushes and electric razors and avoiding activities with injury risks.”
A 2022 study published in the Annals of Internal Medicine found apixaban to be the safest blood thinner among direct oral anticoagulants (DOACs), with the lowest risk of gastrointestinal bleeding.
Excessive and internal bleeding are potential severe risks with blood thinners, which may result in complications ranging from mild bruising to life-threatening hemorrhagic stroke.
When Are Blood Thinners Prescribed
Doctors prescribe blood thinners to decrease the risk of blood clots and for people with irregular heartbeat, heart disease, blood vessel disease, deep vein thrombosis (DVT) and a history of heart attack or stroke.
Your doctor may prescribe a blood thinner if you have certain conditions or experiences.
- Atrial fibrillation (AFib)
- Blood vessel disease, such as deep vein thrombosis (DVT) or pulmonary embolism (PE)
- Congenital heart defects
- Heart disease
- Heart valve replacement surgery
- Post-surgery treatment, when blood clots are a concern
Having AFib comes with high-risk factors, such as rapid heartbeat and increased risk of stroke.
Patients with AFib who took lower doses of blood-thinning medications experienced more bleeding episodes in the first three months of treatment compared to those who took standard doses, according to a 2024 study in the Journal of Blood Advances. Despite these findings, many doctors still prescribe lower doses of these medications to patients with AFib under the assumption that it will reduce bleeding risk.
DVT and PE are two types of venous thromboembolism (VTE), which are blood clots in the veins. DVT happens when a blood clot forms in a deep vein, most often in the legs. PE occurs when a clot forms in one of the pulmonary arteries of the lungs. PE is most often the result of a clot from DVT breaking loose and traveling into the lungs from another body part, such as the arms or legs.
How Do Blood Thinners Work?
Blood thinners work in different ways, such as weakening vitamin K’s blood clotting effects, blocking proteins or enzymes that bind blood cells and platelets or preventing platelets from sticking together or to blood vessel walls.
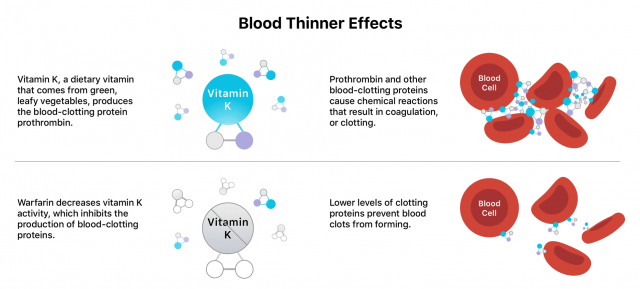
Blood thinners do not actually “thin” the blood or dissolve existing clots, but they can prevent existing clots from worsening.
Types of Blood Thinners
There are two main types of blood thinners: anticoagulants like warfarin and antiplatelet medications like aspirin. They affect blood clotting in different ways.

Anticoagulants
Anticoagulants target and disrupt clotting factors, slowing the body’s process of making clots. Antiplatelet drugs keep platelets from clumping together, preventing a clot from forming. There are various anticoagulants available, with warfarin being one of the oldest and most common options. However, according to a research letter from the JAMA Health Forum, its use is declining. The number of Americans using it has dropped from 2.48 million to just 1.74 million in 2019.
- Warfarin (coumadin)
- It's one of the oldest blood thinners and blocks the formation of vitamin K-dependent clotting factors.
- Eliquis (apixaban)
- This oral medication affects the production of an enzyme, called thrombin, that aids in blood clotting.
- Heparin
- This fast-acting medication is administered via injection or IV. It prevents the production of a fibrin protein that helps the body form clots. Only health care professionals should administer this drug.
- Lovenox (enoxaparin)
- This injectable blood thinner is made from modified heparin, producing a more predictable anticoagulant response.
- Pradaxa (dabigatran) and Xarelto (rivaroxaban)
- These drugs inhibit specific clotting factors in the blood, specifically the enzyme thrombin.
- Savaysa (edoxaban)
- Similar to apixaban, pradaxa and rivaroxaban, it affects thrombin by blocking a vitamin K-dependent protein called Xa.
The FDA has approved several blood thinners since 2010, including Pradaxa, Xarelto, Eliquis and Savaysa. Bayer’s blood thinner candidate, asundexian, faced a setback in a 2023 clinical trial in which the product failed to demonstrate superiority over competing medications. However, asundexian could still become a viable option if its second stroke prevention trial succeeds.
Antiplatelet Drugs
Antiplatelet drugs keep platelets from clumping together, preventing a clot from forming. Aspirin is a widely taken antiplatelet medication recommended for blood clot, heart attack and stroke prevention. It’s available over-the-counter (OTC) and in prescription strength.
Patients typically use the prescription version for rheumatoid arthritis and lupus symptoms. Patients can also combine OTC aspirin with other antiplatelet drugs for acute coronary syndrome or unstable chest pains.
- Low Dose Aspirin
- Often taken by those at higher risk of heart attack or stroke to reduce the risk of another.
- Brilinta (ticagrelor)
- Prescribed for people who’ve survived a heart attack; may be taken with aspirin.
- Effient (prasugrel)
- Effient is often prescribed for individuals who have undergone percutaneous coronary intervention, a procedure that aids in the opening of obstructed coronary (heart) arteries.
- Plavix (clopidogrel)
- Plavix stops blood cells from clumping together by interfering with ADP — a compound that helps blood cells bind together— thus stopping clots and helping blood flow more smoothly.
Doctors rarely prescribe antiplatelet drugs to people who already have blood clots. They instead use them as a preventative medication for those at a higher risk of developing clots.
Natural Blood Thinners
Natural blood thinners like alcohol, garlic, ginger, celery and anise seed can slow clotting, but they are not substitutes for prescription medications. Consult your doctor before using them, as they can have side effects or adverse interactions with prescription blood thinners.
What to Limit When Taking Blood Thinners
Some foods, herbs and oils contain natural blood-thinning properties, which can interfere with blood-thinning medication. Additionally, consuming foods high in vitamin K can impact how blood thinners work, especially warfarin.
It’s important to limit alcohol, drugs and herbal supplements that can counteract blood thinners.
- Alcohol
- Light to moderate alcohol use can increase bleeding risks. Heavy alcohol use can increase the risk of blood clots.
- Antibiotics
- Several antibiotics, such as clarithromycin, ciprofloxacin, metronidazole and trimethoprim, can alter the effectiveness of blood thinners with different levels of risk.
- Antifungal Medications
- These drugs, including ketoconazole or itraconazole, can increase the risk of excess internal bleeding.
- Garlic
- Garlic is a natural blood thinner and may increase the risk of dangerous bleeding when taken with blood thinning medication.
- Ginkgo Biloba
- Like garlic, this herbal supplement also has blood-thinning properties and blood-thinner interactions.
- NSAIDs
- Speak with your doctor before taking non-steroidal anti-inflammatory drugs such as Advil, Aleve, Motrin, Nuprin and Alka-Seltzer.
- St. John’s Wort
- This herbal supplement can decrease the effectiveness of blood thinners.
- Thyroid Medications
- Thyroid drugs can increase the effects of blood thinners.
- Vitamin E
- Some oils, including sunflower, wheat germ, safflower and soybean oil, can increase bleeding risks. Other vitamin-E-rich foods, such as peanut butter, almonds, red bell peppers and pumpkin seeds, can increase bleeding risk as well.
- Vitamin K
- Green leafy foods, including spinach, kale, Swiss chard, parsley, endive, cabbage and asparagus, are high in vitamin K. Avoid vitamin K supplements.
When discussing your diet and medications with your doctor, remember that certain medications may interfere with blood thinners.
- Anti-seizure medications
- Anxiety medications
- Certain antibiotics
- Depression medications
- Fungal medications
- HIV/AIDS medications
- Over-the-counter pain relievers, cold medicines and stomach medications
Blood Thinner Side Effects
The most common side effects of blood thinners are bruising and rash. The most severe side effect is uncontrolled bleeding, especially internal bleeding, which is life-threatening and requires immediate medical attention. According to Austin Chen, a registered pharmacist at Express Scripts, “Patients should… be aware of signs and symptoms of internal bleeding. Slow bleeding may present as fatigue, shortness of breath, pale skin color, and dark, tarry stool. Rapid bleeding can cause stroke symptoms or abdominal and back pain.”
Additionally, Dr. Andrew Youssouf, a board-certified emergency and addiction medicine physician and medical director at Ikon Recovery, told Drugwatch that “Individuals taking blood thinners should be cautious when engaging in physically demanding activities and monitor their intake of foods and supplements that can impact the effectiveness of their medication.”
Call 911 or visit urgent care if you can’t stop the bleeding from a cut. People on blood thinners may experience excessive bleeding from minor cuts and nosebleeds.
- Coughing up blood
- Cuts that won't stop bleeding
- Dark red or dark brown urine
- Excessive bleeding during menstruation
- Nosebleeds or bleeding gums
- Red, dark brown or black stools
- Severe or persistent headaches and stomach pains
Blood thinners can lead to increased bruising, blood blisters, weakness, faintness, dizziness, nausea, aspirin-induced asthma and nasal polyp development.
“My final insights are that patients should always consult their healthcare provider to determine the best course of action for reducing potential risks,” Youssouf said. “Each person’s unique medical history and medication regimen may require different precautions to be taken, so it’s important to discuss with a professional.”
Editor Lindsay Donaldson contributed to this article.
Calling this number connects you with a Drugwatch.com representative. We will direct you to one of our trusted legal partners for a free case review.
Drugwatch.com's trusted legal partners support the organization's mission to keep people safe from dangerous drugs and medical devices. For more information, visit our partners page.

