Smith & Nephew Hip Replacements: Failures, Recalls & Safety Concerns
Smith & Nephew metal-on-metal hip implants have faced serious safety concerns due to design flaws that could cause premature wear and metal debris release. Some patients who received these implants reported severe complications that led to recalls and lawsuits.
Our content is developed and backed by respected legal, medical and scientific experts. More than 30 contributors, including product liability attorneys and board-certified physicians, have reviewed our website to ensure it’s medically sound and legally accurate.
legal help when you need it most.
Drugwatch has provided people injured by harmful drugs and devices with reliable answers and experienced legal help since 2009. Brought to you by The Wilson Firm LLP, we've pursued justice for more than 20,000 families and secured $324 million in settlements and verdicts against negligent manufacturers.
More than 30 contributors, including mass tort attorneys and board-certified doctors, have reviewed our website and added their unique perspectives to ensure you get the most updated and highest quality information.
Drugwatch.com is AACI-certified as a trusted medical content website and is produced by lawyers, a patient advocate and award-winning journalists whose affiliations include the American Bar Association and the American Medical Writers Association.
About Drugwatch.com
- 17 Years of Advocacy
- $324 Million Recovered for Clients
- 20,000 Families Helped
- A+ BBB Rating
- 4.9 Stars from Google Reviews
Testimonials
I found Drugwatch to be very helpful with finding the right lawyers. We had the opportunity to share our story as well, so that more people can be aware of NEC. We are forever grateful for them.
- Legally reviewed by Cassandra L. Sundblad, Esquire
- Last update: March 3, 2026
- Est. Read Time: 5 min read
Why Are Smith & Nephew Hip Implants Linked to Safety Issues?
Smith & Nephew’s metal-on-metal (MoM) hip implants, like the Birmingham Hip Resurfacing and R3 Acetabular System, had serious safety issues due to design flaws.
Over time, they would wear down, potentially releasing harmful metal particles that may cause tissue damage and implant failure. The company recalled some models but faced over 1,000 lawsuits from injured patients.
The Problem With Smith & Nephew Metal-on-Metal Hip Implants
Smith & Nephew’s MoM hip implants contained metal components, usually a ball and socket, that could rub together and wear down. This friction can release harmful metal ions into the bloodstream.
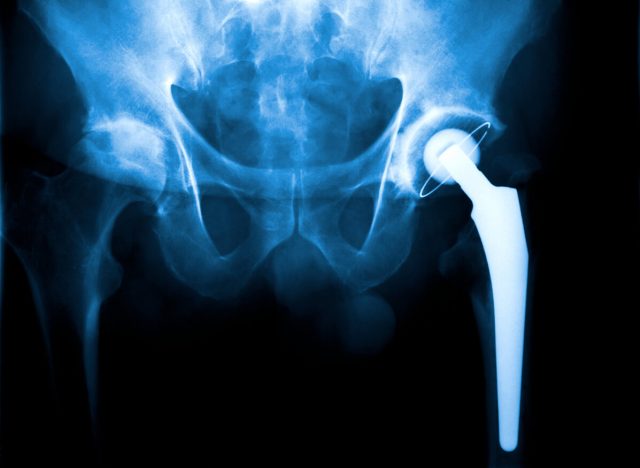
Metal ions released from metal parts rubbing together can destroy bone and soft tissue near the implant and could travel through the bloodstream.
These metal ions sometimes cause metallosis, a form of metal poisoning resulting in inflammation, tissue damage and loose implants. Consequently, many patients needed early replacement surgeries.
After high failure rates were discovered, Smith & Nephew issued recalls and faced lawsuits from patients who claimed the company knew about the risks yet continued to market the implants as safe.
Regulatory & Legal Actions Against Smith & Nephew
Smith & Nephew faced legal issues due to problems with its MoM hip implants, which have high failure rates and serious complications. In 2016, the FDA made regulations stricter, hindering the sale of new MoM implants.
The company recalled several devices over safety concerns and has been involved in hundreds of lawsuits. While most were settled or dismissed, some are still ongoing.
What Has the FDA Done About Smith & Nephew Hip Implant Failures?
The FDA warned Smith & Nephew about quality control issues at its hip implant factory in Tuttlingen, Germany, in December 2010. This warning highlighted failures in testing and correcting problems with their R3 Acetabular System ceramic implants.
In 2016, the U.S. Food and Drug Administration (FDA) made it harder to sell MoM hip implants by requiring manufacturers to get premarket approval. Approval is based on scientific evidence that the device is effective and safe.
“To date, there are no FDA-approved metal-on-metal total hip replacement devices marketed for use in the US. There are two FDA-approved metal-on-metal hip resurfacing devices available.”
New U.S. Food and Drug Administration regulations made it nearly impossible for medical device manufacturers to design, develop or market MoM hips after February 2016. This effectively eliminated MoM hips in the United States.
Smith & Nephew Hip Implant Recalls & Safety Warnings
Smith & Nephew recalled several hip devices between 2012 and 2016 due to high failure rates and reported complications like pain and metallosis.
- Modular SMF and Modular REDAPT Revision Femoral Hip Systems
- On November 15, 2016, Smith & Nephew recalled all Modular SMF and Modular REDAPT Revision Femoral Hip Systems due to increased complaints about their modular necks. The company advised people with the implants to watch for symptoms like pain and swelling. Doctors were told to monitor Cobalt and Chromium metal levels in the blood.
- Birmingham Hip Resurfacing (BHR) system
- In 2015, Smith & Nephew recalled some BHR devices because of high failure rates in people needing smaller sizes. Previously, on July 12, 2007, the company reported a problem with the 56 mm acetabular cup, leading to a Class 2 Device Recall by the FDA.
- R3 Acetabular System
- Overall, the R3 Acetabular System had a higher failure rate than the average for hip replacements. The United Kingdom's National Joint Registry found a 6.3% revision rate within four years, compared to the typical 2.89%. Due to these concerns, the company recalled its metal liner in 2012.
Lawsuits & Settlements Related to Smith & Nephew Hip Implant Failures
Previously, there were almost 1,100 Smith & Nephew BHR lawsuits in a federal multidistrict litigation (MDL). MDLs allow several similar lawsuits to be combined in a single litigation to make the legal process more efficient.
- Misrepresented the safety of the Birmingham Hip Resurfacing device.
- Did not supply the FDA with adverse incident reports.
- Allegedly produced a defective medical device.
- Did not adequately warn doctors and patients about potential risks.
- Provided misleading information regarding the BHR device.
Some Smith & Nephew hip lawsuit claims were dismissed as preempted, but the court allowed discovery on failure to warn, negligence, breach of express warranty and negligent misrepresentation.
In July 2023, Smith & Nephew asked the court to dismiss all lawsuits against the company, but the court refused. Earlier annual reports indicated that there had been “several group, as well as individual, MoM-related settlements without admitting liability.”
As of early 2025, only two cases remain in the MDL, which may indicate that the lawsuits were settled. The Smith & Nephew 2024 Annual Report indicated that about 300 claims against the BHR system are still pending worldwide.
Smith & Nephew’s Role in the Hip Implant Controversy
Smith & Nephew’s BHR system was one of the first MoM hip implants, introduced in 1997. As a pioneer of MoM technology, the company played a significant role in the hip implant controversy of the 2010s.
Initially marketed as a strong and long-lasting option for active individuals, MoM hips were later found to have higher failure rates and serious health concerns, like metal poisoning. Several major manufacturers have faced thousands of combined lawsuits over injuries caused by these devices.
In comparison, DePuy Orthopaedics completely withdrew its Pinnacle implant in 2013 due to similar problems, while Biomet faced lawsuits concerning its M2A Magnum implant but did not recall it.
Smith & Nephew’s response has been less comprehensive, yet its legal challenges are significant.
How Smith & Nephew’s Failures Have Changed the Hip Implant Industry
The failures of Smith & Nephew’s BHR system and other MoM implants have significantly impacted the industry. There are now stricter regulations, improved testing and new technologies to increase safety and implant longevity.
- New Implant Materials
- Smith & Nephew developed OXINIUM, an oxidized zirconium-based material for implants that combines the wear resistance of ceramics with the strength of metal. The company says it resists corrosion better than ceramic and cobalt-chrome materials, addressing concerns with MoM implants.
- Technological Advancements
- Smith & Nephew developed new tools like RI.HIP MODELER and RI.HIP NAVIGATION. These preoperative systems let surgeons improve accuracy and precision when evaluating the relationship and mobility between the spine and pelvis. The company promises these systems reduce the risk of hip dislocation.
These efforts aim to improve implant safety while advancing surgical techniques, making hip replacements more reliable for patients.
What Should You Do if You Have a Smith & Nephew Hip Implant?
Recognizing failure symptoms early is crucial if you have a Smith & Nephew or any MoM hip implant. Regular follow-ups with your orthopedic surgeon can help monitor your implant’s condition and identify potential concerns.
Recognizing Implant Failure & Associated Health Risks
If you have a MoM hip implant, it’s important to follow up with your orthopedic surgeon as recommended. They may suggest more frequent visits depending on your implant type and recovery.
At each appointment, discuss any new or worsening symptoms, like pain, swelling or changes in your ability to walk. Also, update your surgeon on any changes in your overall health.
- Difficulty walking
- Hip or groin pain
- Numbness
- Weakness or swelling
These symptoms might be local, affecting the hip area, but there could also be systemic symptoms involving your skin, kidney, heart, thyroid or nervous system. Metal particles from the implant may cause tissue damage around the joint, leading to loosening and potential implant failure.
In some cases, metal ions enter the bloodstream, causing reactions like:
- Heart problems (cardiomyopathy)
- Kidney issues
- Neurological changes, like hearing or vision impairments
- Psychological issues, including depression or cognitive decline
- Skin rashes
- Thyroid problems, like fatigue and weight gain
It’s important to consult your doctor if you experience any of these symptoms.
Resources for People With a Smith & Nephew Hip Implant
If you had hip replacement surgery before May 18, 2016, you might have a MoM implant, which can pose risks over time. The FDA suggests consulting your doctor for information on your implant type and safety concerns.
- Arthritis Foundation Metal-on-Metal Hip Implant Risks Page
- This resource from the Arthritis Foundation provides details on the potential risks of MoM hip implants and assists patients facing complications with these devices.
- Device Recall Dashboard
- This online search engine from the American Academy of Orthopaedic Surgeons (AAOS) allows users to search for specific brands of joint implants to see if they have been recalled.
- FDA Metal-on-Metal Hip Implants Information Page
- This FDA webpage offers comprehensive information on MoM hip implants, including potential risks, guidance for patients and current safety updates.
- Massachusetts General Hospital (Mass General)
- The Mass General Center for Metal-on-Metal Hip & Tribocorrosion Evaluation & Treatment addresses issues with metal-on-metal hip implants, ensuring patients receive optimal care.
- MedWatch
- This FDA system lets you report any adverse reactions possibly linked to your MoM hip implant.
Calling this number connects you with a Drugwatch.com representative. We will direct you to one of our trusted legal partners for a free case review.
Drugwatch.com's trusted legal partners support the organization's mission to keep people safe from dangerous drugs and medical devices. For more information, visit our partners page.


