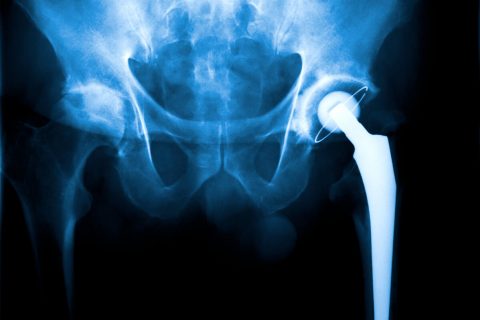
Wright Medical Hip Implant Lawsuits
Wright Medical agreed to two settlements totaling $330 million to resolve hundreds of lawsuits over its hip-replacement implants. After the final settlement, a federal court closed the multidistrict litigation against Wright in June 2018. The company no longer makes hip implants.
Our content is developed and backed by respected legal, medical and scientific experts. More than 30 contributors, including product liability attorneys and board-certified physicians, have reviewed our website to ensure it’s medically sound and legally accurate.
legal help when you need it most.
Drugwatch has provided people injured by harmful drugs and devices with reliable answers and experienced legal help since 2009. Brought to you by The Wilson Firm LLP, we've pursued justice for more than 20,000 families and secured $324 million in settlements and verdicts against negligent manufacturers.
More than 30 contributors, including mass tort attorneys and board-certified doctors, have reviewed our website and added their unique perspectives to ensure you get the most updated and highest quality information.
Drugwatch.com is AACI-certified as a trusted medical content website and is produced by lawyers, a patient advocate and award-winning journalists whose affiliations include the American Bar Association and the American Medical Writers Association.
About Drugwatch.com
- 17 Years of Advocacy
- $324 Million Recovered for Clients
- 20,000 Families Helped
- A+ BBB Rating
- 4.9 Stars from Google Reviews
Testimonials
I found Drugwatch to be very helpful with finding the right lawyers. We had the opportunity to share our story as well, so that more people can be aware of NEC. We are forever grateful for them.
- Legally reviewed by Whitney Ray Di Bona, Esquire
- Last update: April 1, 2026
- Est. Read Time: 3 min read
Latest Updates in Wright Medical Hip Implant Lawsuits
Some lawyers are still accepting individual hip implant cases, but the multidistrict litigation is closed. After losing its first two bellwether test trials, Wright Medical offered to settle its remaining hip implant lawsuits.
Wright Medical faced roughly 2,000 lawsuits after selling its hip implant manufacturing operations. A federal panel combined 640 of the lawsuits into a multidistrict litigation in Georgia. Others were combined in state courts.
- June 2026: The MDL has already been closed for years, but some lawyers were still evaluating individual cases for potential settlement negotiations or to go to trial.
- December 2023: Profemur brand artificial hip implants have broken apart at the dual modular “neck,” causing patients to suddenly lose their ability to walk or stand, according to a joint investigation by KFF Health and CBS News.
- September 2019: Final settlement payments from Wright were supposed to be sent out.
- July 2019: 73 lawsuits remained in the MDL.
- June 2018: The court ordered the MDL closed.
- October 2017: Wright agreed to settle all remaining hip-implant lawsuits against the company — about 600 cases — for $90 million. No additional lawsuits could be filed in the MDL.
- November 2016: Wright settled more than 1,200 lawsuits for $240 million.
The second settlement in October 2017 covered plaintiffs who had not been allowed into the first settlement. This included people who filed lawsuits after the November 2016 agreement and those who had missed the deadline to file a lawsuit.
The 2017 settlement also applied to dozens of cases that were still pending in a Judicial Council Coordination Proceeding in California state court. No additional lawsuits could be added to the MDL after Oct. 18, 2017. Following the 2017 settlement, the court ordered the MDL closed in June 2018.
Wright Hip Implants Named in Lawsuits
Wright’s hip implants were manufactured by the company’s OrthoRecon division. Wright sold OrthoRecon in 2013 but remained legally responsible for lawsuits over its hips. Wright Medical no longer manufactures hip implants.
Wright hips named in lawsuits:
- Conserve Total Hip Implant System
- Conserve Total A-Class Advanced Metal Hip Implant System
- Conserve Resurfacing System
- Profemur System
- Dynasty hip implants
- Lineage hip implants
The majority of lawsuits named Wright’s Conserve and Profemur hip devices. The MDL was limited to only Conserve hips and Profemur System parts related to certain Conserve components.
Hip Failure Rate Higher than Expected
Studies and reports to federal regulators have found many Wright hip implants failed sooner or more often than expected.
A 2016 review published in The Open Orthopaedics Journal found a high failure rate among patients who received Wright Conserve hips. The study looked at 92 patients who received a Conserve hip between 2005 and 2010. It found that nearly 1 in every 5 patients needed revision surgery to correct problems with the devices. The average time between receiving a Wright hip replacement and requiring corrective surgery was just 4 1/2 years.
In 2015, the company that had purchased Wright Medical’s hip division started receiving a higher-than-expected number of reports of fractures with the Profemur. MicroPort Orthopedics recalled 10,825 of the devices.
“If the modular neck fractures, the patient may experience sudden pain, instability and difficulty walking and performing common task.”
The FDA declared the Profemur recall a Class I recall, the agency’s most serious type. The recall notice warned that an acute fracture of the device would require emergency surgery. It also warned that such a fracture in a Profemur hip stem could “lead to neurovascular damage, hematoma, hemorrhage, and even death.
People filed their lawsuits based on the failure of these hips and the complications that occurred as a result.
- John Wallace
- John Wallace of Arizona received a Conserve implant in his left hip in 2005. In December 2011, his doctor found elevated chromium and cobalt levels in his blood. Wallace required revision surgery which led to further complications including a “spontaneous dislocation” of the hip.
- Linda Bohnenstiehl
- Linda Bohnenstiehl of Illinois received Conserve and Profemur hip devices in a Missouri hospital in 2008. She claimed that the Profemur device fractured at the femoral neck in 2010, causing her physical injuries, debilitation and pain and suffering. The injuries forced her to have a revision surgery.
- Leon Thomas
- Leon Thomas of Arkansas received a Conserve Total Hip System in 2006. He began experiencing severe pain in his left hip and groin in 2011 and was forced to have revision surgery. His doctor suspected a loose hip socket, and he found elevated chromium and cobalt levels in his blood. Thomas’ lawsuit claimed he was permanently impaired and suffered ongoing discomfort from nerve damage and other injuries.
Juries Award $15.5 Million, Wright Offers $330 Million in Settlements
Two juries returned verdicts totaling $15.5 million against Wright in lawsuits over the company’s hip replacements. The back-to-back losses appear to have motivated the company to offer up $330 million through two settlements to resolve the remaining cases.
-
November 2013
Wright settled the first lawsuit over its Profemur hip stem for an undisclosed amount five days before trial. Plaintiff Timothy Courson had claimed permanent injuries and had sought $3 million in damages.
-
June 2015
A California jury awarded Alan Warner $4.5 million in the first Wright hip lawsuit to go to trial. Warner claimed the Profemur device snapped while he was walking. The court later reduced the award to $1 million.
-
November 2015
Jury awarded $11 million to former ski instructor Robyn Christiansen after finding a Conserve implant caused tissue damage. The court later reduced the amount to $2.1 million.
-
November 2016
Wright settled 1,292 lawsuits in the federal MDL and the consolidated court action in California for a total of $240 million.
-
October 2017
Wright agreed to settle roughly 600 remaining hip lawsuits for $90 million.
-
June 2018
A judge closed the Wright MDL, barring new lawsuits.
-
September 2019
Wright estimated it would make the final payment to patients by this date.
Calling this number connects you with a Drugwatch.com representative. We will direct you to one of our trusted legal partners for a free case review.
Drugwatch.com's trusted legal partners support the organization's mission to keep people safe from dangerous drugs and medical devices. For more information, visit our partners page.







